| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Plasminogen activator inhibitor 1 |
|---|
| Ligand | BDBM50111273 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_222792 (CHEMBL847120) |
|---|
| IC50 | 290±n/a nM |
|---|
| Citation |  Folkes, A; Brown, SD; Canne, LE; Chan, J; Engelhardt, E; Epshteyn, S; Faint, R; Golec, J; Hanel, A; Kearney, P; Leahy, JW; Mac, M; Matthews, D; Prisbylla, MP; Sanderson, J; Simon, RJ; Tesfai, Z; Vicker, N; Wang, S; Webb, RR; Charlton, P Design, synthesis and in vitro evaluation of potent, novel, small molecule inhibitors of plasminogen activator inhibitor-1. Bioorg Med Chem Lett12:1063-6 (2002) [PubMed] Folkes, A; Brown, SD; Canne, LE; Chan, J; Engelhardt, E; Epshteyn, S; Faint, R; Golec, J; Hanel, A; Kearney, P; Leahy, JW; Mac, M; Matthews, D; Prisbylla, MP; Sanderson, J; Simon, RJ; Tesfai, Z; Vicker, N; Wang, S; Webb, RR; Charlton, P Design, synthesis and in vitro evaluation of potent, novel, small molecule inhibitors of plasminogen activator inhibitor-1. Bioorg Med Chem Lett12:1063-6 (2002) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Plasminogen activator inhibitor 1 |
|---|
| Name: | Plasminogen activator inhibitor 1 |
|---|
| Synonyms: | Endothelial plasminogen activator inhibitor | PAI | PAI-1 | PAI1 | PAI1_HUMAN | PLANH1 | Plasminogen activator inhibitor 1 | Plasminogen activator inhibitor 1 (PAI-1) | Plasminogen activator inhibitor-1 | Plasminogen activator-1 (PAI-1) | SERPINE1 |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 45064.00 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P05121 |
|---|
| Residue: | 402 |
|---|
| Sequence: | MQMSPALTCLVLGLALVFGEGSAVHHPPSYVAHLASDFGVRVFQQVAQASKDRNVVFSPY
GVASVLAMLQLTTGGETQQQIQAAMGFKIDDKGMAPALRHLYKELMGPWNKDEISTTDAI
FVQRDLKLVQGFMPHFFRLFRSTVKQVDFSEVERARFIINDWVKTHTKGMISNLLGKGAV
DQLTRLVLVNALYFNGQWKTPFPDSSTHRRLFHKSDGSTVSVPMMAQTNKFNYTEFTTPD
GHYYDILELPYHGDTLSMFIAAPYEKEVPLSALTNILSAQLISHWKGNMTRLPRLLVLPK
FSLETEVDLRKPLENLGMTDMFRQFQADFTSLSDQEPLHVAQALQKVKIEVNESGTVASS
STAVIVSARMAPEEIIMDRPFLFVVRHNPTGTVLFMGQVMEP
|
|
|
|---|
| BDBM50111273 |
|---|
| n/a |
|---|
| Name | BDBM50111273 |
|---|
| Synonyms: | 6-Benzo[b]thiophen-2-yl-4-hydroxy-2-oxo-1,2-dihydro-quinoline-3-carboxylic acid [4-(7-tetrazol-1-yl-heptyloxy)-phenyl]-amide | CHEMBL10004 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C32H30N6O4S |
|---|
| Mol. Mass. | 594.683 |
|---|
| SMILES | Oc1c(C(=O)Nc2ccc(OCCCCCCCn3cnnn3)cc2)c(=O)[nH]c2ccc(cc12)-c1cc2ccccc2s1 |
|---|
| Structure | 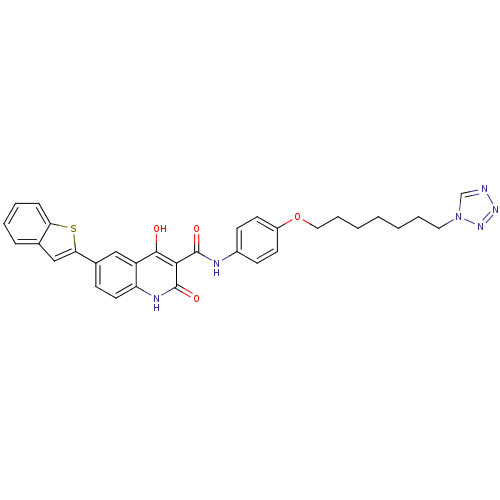
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Folkes, A; Brown, SD; Canne, LE; Chan, J; Engelhardt, E; Epshteyn, S; Faint, R; Golec, J; Hanel, A; Kearney, P; Leahy, JW; Mac, M; Matthews, D; Prisbylla, MP; Sanderson, J; Simon, RJ; Tesfai, Z; Vicker, N; Wang, S; Webb, RR; Charlton, P Design, synthesis and in vitro evaluation of potent, novel, small molecule inhibitors of plasminogen activator inhibitor-1. Bioorg Med Chem Lett12:1063-6 (2002) [PubMed]
Folkes, A; Brown, SD; Canne, LE; Chan, J; Engelhardt, E; Epshteyn, S; Faint, R; Golec, J; Hanel, A; Kearney, P; Leahy, JW; Mac, M; Matthews, D; Prisbylla, MP; Sanderson, J; Simon, RJ; Tesfai, Z; Vicker, N; Wang, S; Webb, RR; Charlton, P Design, synthesis and in vitro evaluation of potent, novel, small molecule inhibitors of plasminogen activator inhibitor-1. Bioorg Med Chem Lett12:1063-6 (2002) [PubMed]