| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Methionine--tRNA ligase, cytoplasmic |
|---|
| Ligand | BDBM50112575 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_105280 (CHEMBL718948) |
|---|
| IC50 | 1±n/a nM |
|---|
| Citation |  Jarvest, RL; Berge, JM; Berry, V; Boyd, HF; Brown, MJ; Elder, JS; Forrest, AK; Fosberry, AP; Gentry, DR; Hibbs, MJ; Jaworski, DD; O'Hanlon, PJ; Pope, AJ; Rittenhouse, S; Sheppard, RJ; Slater-Radosti, C; Worby, A Nanomolar inhibitors of Staphylococcus aureus methionyl tRNA synthetase with potent antibacterial activity against gram-positive pathogens. J Med Chem45:1959-62 (2002) [PubMed] Jarvest, RL; Berge, JM; Berry, V; Boyd, HF; Brown, MJ; Elder, JS; Forrest, AK; Fosberry, AP; Gentry, DR; Hibbs, MJ; Jaworski, DD; O'Hanlon, PJ; Pope, AJ; Rittenhouse, S; Sheppard, RJ; Slater-Radosti, C; Worby, A Nanomolar inhibitors of Staphylococcus aureus methionyl tRNA synthetase with potent antibacterial activity against gram-positive pathogens. J Med Chem45:1959-62 (2002) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Methionine--tRNA ligase, cytoplasmic |
|---|
| Name: | Methionine--tRNA ligase, cytoplasmic |
|---|
| Synonyms: | MARS | MARS1 | Methionyl-tRNA synthetase | SYMC_HUMAN |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 101110.04 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | ChEMBL_105283 |
|---|
| Residue: | 900 |
|---|
| Sequence: | MRLFVSDGVPGCLPVLAAAGRARGRAEVLISTVGPEDCVVPFLTRPKVPVLQLDSGNYLF
STSAICRYFFLLSGWEQDDLTNQWLEWEATELQPALSAALYYLVVQGKKGEDVLGSVRRA
LTHIDHSLSRQNCPFLAGETESLADIVLWGALYPLLQDPAYLPEELSALHSWFQTLSTQE
PCQRAAETVLKQQGVLALRPYLQKQPQPSPAEGRAVTNEPEEEELATLSEEEIAMAVTAW
EKGLESLPPLRPQQNPVLPVAGERNVLITSALPYVNNVPHLGNIIGCVLSADVFARYSRL
RQWNTLYLCGTDEYGTATETKALEEGLTPQEICDKYHIIHADIYRWFNISFDIFGRTTTP
QQTKITQDIFQQLLKRGFVLQDTVEQLRCEHCARFLADRFVEGVCPFCGYEEARGDQCDK
CGKLINAVELKKPQCKVCRSCPVVQSSQHLFLDLPKLEKRLEEWLGRTLPGSDWTPNAQF
ITRSWLRDGLKPRCITRDLKWGTPVPLEGFEDKVFYVWFDATIGYLSITANYTDQWERWW
KNPEQVDLYQFMAKDNVPFHSLVFPCSALGAEDNYTLVSHLIATEYLNYEDGKFSKSRGV
GVFGDMAQDTGIPADIWRFYLLYIRPEGQDSAFSWTDLLLKNNSELLNNLGNFINRAGMF
VSKFFGGYVPEMVLTPDDQRLLAHVTLELQHYHQLLEKVRIRDALRSILTISRHGNQYIQ
VNEPWKRIKGSEADRQRAGTVTGLAVNIAALLSVMLQPYMPTVSATIQAQLQLPPPACSI
LLTNFLCTLPAGHQIGTVSPLFQKLENDQIESLRQRFGGGQAKTSPKPAVVETVTTAKPQ
QIQALMDEVTKQGNIVRELKAQKADKNEVAAEVAKLLDLKKQLAVAEGKPPEAPKGKKKK
|
|
|
|---|
| BDBM50112575 |
|---|
| n/a |
|---|
| Name | BDBM50112575 |
|---|
| Synonyms: | 2-[3-(6,8-Dibromo-1,2,3,4-tetrahydro-quinolin-4-ylamino)-propylamino]-1H-quinolin-4-one | CHEMBL304237 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C21H22Br2N4O |
|---|
| Mol. Mass. | 506.234 |
|---|
| SMILES | Oc1cc(NCCCNC2CCNc3c(Br)cc(Br)cc23)nc2ccccc12 |
|---|
| Structure | 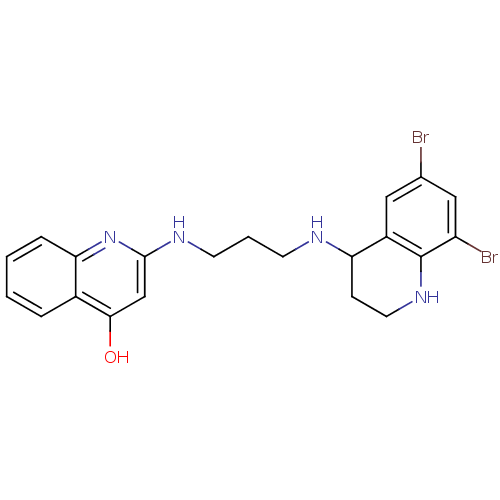
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Jarvest, RL; Berge, JM; Berry, V; Boyd, HF; Brown, MJ; Elder, JS; Forrest, AK; Fosberry, AP; Gentry, DR; Hibbs, MJ; Jaworski, DD; O'Hanlon, PJ; Pope, AJ; Rittenhouse, S; Sheppard, RJ; Slater-Radosti, C; Worby, A Nanomolar inhibitors of Staphylococcus aureus methionyl tRNA synthetase with potent antibacterial activity against gram-positive pathogens. J Med Chem45:1959-62 (2002) [PubMed]
Jarvest, RL; Berge, JM; Berry, V; Boyd, HF; Brown, MJ; Elder, JS; Forrest, AK; Fosberry, AP; Gentry, DR; Hibbs, MJ; Jaworski, DD; O'Hanlon, PJ; Pope, AJ; Rittenhouse, S; Sheppard, RJ; Slater-Radosti, C; Worby, A Nanomolar inhibitors of Staphylococcus aureus methionyl tRNA synthetase with potent antibacterial activity against gram-positive pathogens. J Med Chem45:1959-62 (2002) [PubMed]