| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Type-1 angiotensin II receptor B |
|---|
| Ligand | BDBM50091105 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_34836 (CHEMBL648770) |
|---|
| Ki | >0.000100±n/a nM |
|---|
| Citation |  Murugesan, N; Tellew, JE; Gu, Z; Kunst, BL; Fadnis, L; Cornelius, LA; Baska, RA; Yang, Y; Beyer, SM; Monshizadegan, H; Dickinson, KE; Panchal, B; Valentine, MT; Chong, S; Morrison, RA; Carlson, KE; Powell, JR; Moreland, S; Barrish, JC; Kowala, MC; Macor, JE Discovery of N-isoxazolyl biphenylsulfonamides as potent dual angiotensin II and endothelin A receptor antagonists. J Med Chem45:3829-35 (2002) [PubMed] Murugesan, N; Tellew, JE; Gu, Z; Kunst, BL; Fadnis, L; Cornelius, LA; Baska, RA; Yang, Y; Beyer, SM; Monshizadegan, H; Dickinson, KE; Panchal, B; Valentine, MT; Chong, S; Morrison, RA; Carlson, KE; Powell, JR; Moreland, S; Barrish, JC; Kowala, MC; Macor, JE Discovery of N-isoxazolyl biphenylsulfonamides as potent dual angiotensin II and endothelin A receptor antagonists. J Med Chem45:3829-35 (2002) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Type-1 angiotensin II receptor B |
|---|
| Name: | Type-1 angiotensin II receptor B |
|---|
| Synonyms: | AGTRB_RAT | AT3 | Agtr1 | Agtr1b | Angiotensin II AT1B | Angiotensin II receptor (AT-1) type-1 | Angiotensin II type 1b (AT-1b) receptor | At1b | Type-1B angiotensin II receptor |
|---|
| Type: | Enzyme Catalytic Domain |
|---|
| Mol. Mass.: | 40929.44 |
|---|
| Organism: | RAT |
|---|
| Description: | Angiotensin II AT1B 0 RAT::P29089 |
|---|
| Residue: | 359 |
|---|
| Sequence: | MTLNSSTEDGIKRIQDDCPKAGRHNYIFVMIPTLYSIIFVVGIFGNSLVVIVIYFYMKLK
TVASVFLLNLALADLCFLLTLPLWAVYTAMEYRWPFGNHLCKIASASVSFNLYASVFLLT
CLSIDRYLAIVHPMKSRLRRTMLVAKVTCIIIWLMAGLASLPAVIYRNVYFIENTNITVC
AFHYESQNSTLPIGLGLTKNILGFVFPFLIILTSYTLIWKALKKAYKIQKNTPRNDDIFR
IIMAIVLFFFFSWVPHQIFTFLDVLIQLGIIRDCEIADIVDTAMPITICIAYFNNCLNPL
FYGFLGKKFKKYFLQLLKYIPPTAKSHAGLSTKMSTLSYRPSDNMSSSAKKSASFFEVE
|
|
|
|---|
| BDBM50091105 |
|---|
| n/a |
|---|
| Name | BDBM50091105 |
|---|
| Synonyms: | 4''-Oxazol-2-yl-biphenyl-2-sulfonic acid (3,4-dimethyl-isoxazol-5-yl)-amide | 4'-Oxazol-2-yl-biphenyl-2-sulfonic acid (3,4-dimethyl-isoxazol-5-yl)-amide | 4'-Oxazol-2-yl-biphenyl-2-sulfonicacid(3,4-dimethyl-isoxazol-5-yl)-amide | BMS-193884 | CHEMBL24461 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C20H17N3O4S |
|---|
| Mol. Mass. | 395.432 |
|---|
| SMILES | Cc1noc(NS(=O)(=O)c2ccccc2-c2ccc(cc2)-c2ncco2)c1C |
|---|
| Structure | 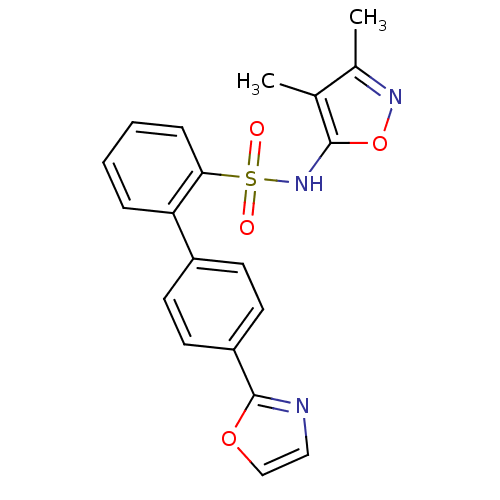
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Murugesan, N; Tellew, JE; Gu, Z; Kunst, BL; Fadnis, L; Cornelius, LA; Baska, RA; Yang, Y; Beyer, SM; Monshizadegan, H; Dickinson, KE; Panchal, B; Valentine, MT; Chong, S; Morrison, RA; Carlson, KE; Powell, JR; Moreland, S; Barrish, JC; Kowala, MC; Macor, JE Discovery of N-isoxazolyl biphenylsulfonamides as potent dual angiotensin II and endothelin A receptor antagonists. J Med Chem45:3829-35 (2002) [PubMed]
Murugesan, N; Tellew, JE; Gu, Z; Kunst, BL; Fadnis, L; Cornelius, LA; Baska, RA; Yang, Y; Beyer, SM; Monshizadegan, H; Dickinson, KE; Panchal, B; Valentine, MT; Chong, S; Morrison, RA; Carlson, KE; Powell, JR; Moreland, S; Barrish, JC; Kowala, MC; Macor, JE Discovery of N-isoxazolyl biphenylsulfonamides as potent dual angiotensin II and endothelin A receptor antagonists. J Med Chem45:3829-35 (2002) [PubMed]