| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Endothelin-converting enzyme 1 |
|---|
| Ligand | BDBM50119746 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEBML_64356 |
|---|
| IC50 | 23±n/a nM |
|---|
| Citation |  Firooznia, F; Gude, C; Chan, K; Tan, J; Fink, CA; Savage, P; Beil, ME; Bruseo, CW; Trapani, AJ; Jeng, AY Synthesis and biological activity of potent heterocyclic thiol-based inhibitors of endothelin-converting enzyme-1. Bioorg Med Chem Lett12:3059-62 (2002) [PubMed] Firooznia, F; Gude, C; Chan, K; Tan, J; Fink, CA; Savage, P; Beil, ME; Bruseo, CW; Trapani, AJ; Jeng, AY Synthesis and biological activity of potent heterocyclic thiol-based inhibitors of endothelin-converting enzyme-1. Bioorg Med Chem Lett12:3059-62 (2002) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Endothelin-converting enzyme 1 |
|---|
| Name: | Endothelin-converting enzyme 1 |
|---|
| Synonyms: | ECE-1 | ECE1 | ECE1_HUMAN | Endothelin-Converting Enzyme 1 | Endothelin-converting enzyme 1 (ECE1) |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 87155.11 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P42892 |
|---|
| Residue: | 770 |
|---|
| Sequence: | MRGVWPPPVSALLSALGMSTYKRATLDEEDLVDSLSEGDAYPNGLQVNFHSPRSGQRCWA
ARTQVEKRLVVLVVLLAAGLVACLAALGIQYQTRSPSVCLSEACVSVTSSILSSMDPTVD
PCHDFFSYACGGWIKANPVPDGHSRWGTFSNLWEHNQAIIKHLLENSTASVSEAERKAQV
YYRACMNETRIEELRAKPLMELIERLGGWNITGPWAKDNFQDTLQVVTAHYRTSPFFSVY
VSADSKNSNSNVIQVDQSGLGLPSRDYYLNKTENEKVLTGYLNYMVQLGKLLGGGDEEAI
RPQMQQILDFETALANITIPQEKRRDEELIYHKVTAAELQTLAPAINWLPFLNTIFYPVE
INESEPIVVYDKEYLEQISTLINTTDRCLLNNYMIWNLVRKTSSFLDQRFQDADEKFMEV
MYGTKKTCLPRWKFCVSDTENNLGFALGPMFVKATFAEDSKSIATEIILEIKKAFEESLS
TLKWMDEETRKSAKEKADAIYNMIGYPNFIMDPKELDKVFNDYTAVPDLYFENAMRFFNF
SWRVTADQLRKAPNRDQWSMTPPMVNAYYSPTKNEIVFPAGILQAPFYTRSSPKALNFGG
IGVVVGHELTHAFDDQGREYDKDGNLRPWWKNSSVEAFKRQTECMVEQYSNYSVNGEPVN
GRHTLGENIADNGGLKAAYRAYQNWVKKNGAEHSLPTLGLTNNQLFFLGFAQVWCSVRTP
ESSHEGLITDPHSPSRFRVIGSLSNSKEFSEHFRCPPGSPMNPPHKCEVW
|
|
|
|---|
| BDBM50119746 |
|---|
| n/a |
|---|
| Name | BDBM50119746 |
|---|
| Synonyms: | 2-{[1-((S)-3-Cyclohexyl-2-mercapto-propionylamino)-cyclopentanecarbonyl]-amino}-3-(6-thiophen-3-yl-pyridin-3-yl)-propionic acid | CHEMBL319463 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C27H35N3O4S2 |
|---|
| Mol. Mass. | 529.715 |
|---|
| SMILES | OC(=O)C(Cc1ccc(nc1)-c1ccsc1)NC(=O)C1(CCCC1)NC(=O)[C@@H](S)CC1CCCCC1 |
|---|
| Structure | 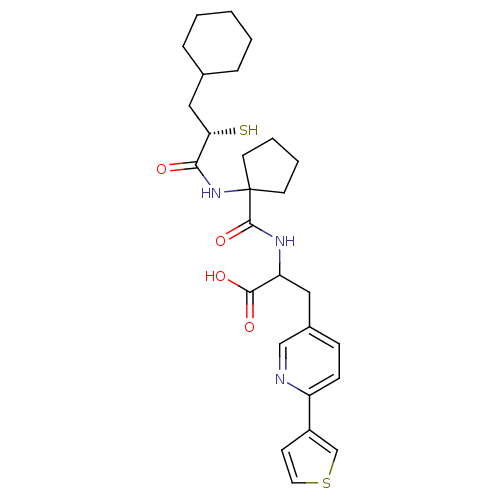
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Firooznia, F; Gude, C; Chan, K; Tan, J; Fink, CA; Savage, P; Beil, ME; Bruseo, CW; Trapani, AJ; Jeng, AY Synthesis and biological activity of potent heterocyclic thiol-based inhibitors of endothelin-converting enzyme-1. Bioorg Med Chem Lett12:3059-62 (2002) [PubMed]
Firooznia, F; Gude, C; Chan, K; Tan, J; Fink, CA; Savage, P; Beil, ME; Bruseo, CW; Trapani, AJ; Jeng, AY Synthesis and biological activity of potent heterocyclic thiol-based inhibitors of endothelin-converting enzyme-1. Bioorg Med Chem Lett12:3059-62 (2002) [PubMed]