| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Mitogen-activated protein kinase 14 |
|---|
| Ligand | BDBM50122401 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEBML_124486 |
|---|
| IC50 | 1.5±n/a nM |
|---|
| Citation |  Stelmach, JE; Liu, L; Patel, SB; Pivnichny, JV; Scapin, G; Singh, S; Hop, CE; Wang, Z; Strauss, JR; Cameron, PM; Nichols, EA; O'Keefe, SJ; O'Neill, EA; Schmatz, DM; Schwartz, CD; Thompson, CM; Zaller, DM; Doherty, JB Design and synthesis of potent, orally bioavailable dihydroquinazolinone inhibitors of p38 MAP kinase. Bioorg Med Chem Lett13:277-80 (2002) [PubMed] Stelmach, JE; Liu, L; Patel, SB; Pivnichny, JV; Scapin, G; Singh, S; Hop, CE; Wang, Z; Strauss, JR; Cameron, PM; Nichols, EA; O'Keefe, SJ; O'Neill, EA; Schmatz, DM; Schwartz, CD; Thompson, CM; Zaller, DM; Doherty, JB Design and synthesis of potent, orally bioavailable dihydroquinazolinone inhibitors of p38 MAP kinase. Bioorg Med Chem Lett13:277-80 (2002) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Mitogen-activated protein kinase 14 |
|---|
| Name: | Mitogen-activated protein kinase 14 |
|---|
| Synonyms: | CSAID-binding protein | CSBP | CSBP1 | CSBP2 | CSPB1 | Cytokine suppressive anti-inflammatory drug-binding protein | MAP kinase 14 | MAP kinase MXI2 | MAP kinase p38 alpha | MAPK 14 | MAPK14 | MAX-interacting protein 2 | MK14_HUMAN | MXI2 | Mitogen-activated protein kinase p38 alpha | SAPK2A | Stress-activated protein kinase 2a | p38 MAP kinase alpha/beta |
|---|
| Type: | Serine/threonine-protein kinase |
|---|
| Mol. Mass.: | 41286.76 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Q16539 |
|---|
| Residue: | 360 |
|---|
| Sequence: | MSQERPTFYRQELNKTIWEVPERYQNLSPVGSGAYGSVCAAFDTKTGLRVAVKKLSRPFQ
SIIHAKRTYRELRLLKHMKHENVIGLLDVFTPARSLEEFNDVYLVTHLMGADLNNIVKCQ
KLTDDHVQFLIYQILRGLKYIHSADIIHRDLKPSNLAVNEDCELKILDFGLARHTDDEMT
GYVATRWYRAPEIMLNWMHYNQTVDIWSVGCIMAELLTGRTLFPGTDHIDQLKLILRLVG
TPGAELLKKISSESARNYIQSLTQMPKMNFANVFIGANPLAVDLLEKMLVLDSDKRITAA
QALAHAYFAQYHDPDDEPVADPYDQSFESRDLLIDEWKSLTYDEVISFVPPPLDQEEMES
|
|
|
|---|
| BDBM50122401 |
|---|
| n/a |
|---|
| Name | BDBM50122401 |
|---|
| Synonyms: | 5-(2-Chloro-phenyl)-1-(2,6-dichloro-phenyl)-7-(piperazine-1-carbonyl)-3,4-dihydro-1H-quinazolin-2-one | 5-(2-chlorophenyl)-1-(2,6-dichlorophenyl)-7-(piperazine-1-carbonyl)-3,4-dihydroquinazolin-2(1H)-one | CHEMBL94444 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C25H21Cl3N4O2 |
|---|
| Mol. Mass. | 515.819 |
|---|
| SMILES | Clc1ccccc1-c1cc(cc2N(C(=O)NCc12)c1c(Cl)cccc1Cl)C(=O)N1CCNCC1 |
|---|
| Structure | 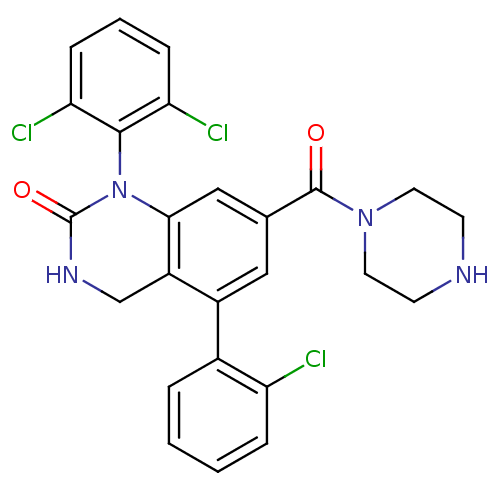
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Stelmach, JE; Liu, L; Patel, SB; Pivnichny, JV; Scapin, G; Singh, S; Hop, CE; Wang, Z; Strauss, JR; Cameron, PM; Nichols, EA; O'Keefe, SJ; O'Neill, EA; Schmatz, DM; Schwartz, CD; Thompson, CM; Zaller, DM; Doherty, JB Design and synthesis of potent, orally bioavailable dihydroquinazolinone inhibitors of p38 MAP kinase. Bioorg Med Chem Lett13:277-80 (2002) [PubMed]
Stelmach, JE; Liu, L; Patel, SB; Pivnichny, JV; Scapin, G; Singh, S; Hop, CE; Wang, Z; Strauss, JR; Cameron, PM; Nichols, EA; O'Keefe, SJ; O'Neill, EA; Schmatz, DM; Schwartz, CD; Thompson, CM; Zaller, DM; Doherty, JB Design and synthesis of potent, orally bioavailable dihydroquinazolinone inhibitors of p38 MAP kinase. Bioorg Med Chem Lett13:277-80 (2002) [PubMed]