| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Mitogen-activated protein kinase 14 |
|---|
| Ligand | BDBM50122915 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_124502 (CHEMBL733338) |
|---|
| IC50 | 8±n/a nM |
|---|
| Citation |  Colletti, SL; Frie, JL; Dixon, EC; Singh, SB; Choi, BK; Scapin, G; Fitzgerald, CE; Kumar, S; Nichols, EA; O'Keefe, SJ; O'Neill, EA; Porter, G; Samuel, K; Schmatz, DM; Schwartz, CD; Shoop, WL; Thompson, CM; Thompson, JE; Wang, R; Woods, A; Zaller, DM; Doherty, JB Hybrid-designed inhibitors of p38 MAP kinase utilizing N-arylpyridazinones. J Med Chem46:349-52 (2003) [PubMed] Article Colletti, SL; Frie, JL; Dixon, EC; Singh, SB; Choi, BK; Scapin, G; Fitzgerald, CE; Kumar, S; Nichols, EA; O'Keefe, SJ; O'Neill, EA; Porter, G; Samuel, K; Schmatz, DM; Schwartz, CD; Shoop, WL; Thompson, CM; Thompson, JE; Wang, R; Woods, A; Zaller, DM; Doherty, JB Hybrid-designed inhibitors of p38 MAP kinase utilizing N-arylpyridazinones. J Med Chem46:349-52 (2003) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Mitogen-activated protein kinase 14 |
|---|
| Name: | Mitogen-activated protein kinase 14 |
|---|
| Synonyms: | Crk1 | Csbp1 | Csbp2 | MAP Kinase p38 alpha | MAP kinase p38 | MK14_MOUSE | Mapk14 | Mitogen-activated protein kinase 14 | Mitogen-activated protein kinase p38 alpha |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 41281.22 |
|---|
| Organism: | Mus musculus (mouse) |
|---|
| Description: | The full-length open reading frame of murine p38 alpha was cloned and expressed in E. coli.. Soluble murine p38R was extracted from cell pellets and purified using ion-exchange chromatography. |
|---|
| Residue: | 360 |
|---|
| Sequence: | MSQERPTFYRQELNKTIWEVPERYQNLSPVGSGAYGSVCAAFDTKTGHRVAVKKLSRPFQ
SIIHAKRTYRELRLLKHMKHENVIGLLDVFTPARSLEEFNDVYLVTHLMGADLNNIVKCQ
KLTDDHVQFLIYQILRGLKYIHSADIIHRDLKPSNLAVNEDCELKILDFGLARHTDDEMT
GYVATRWYRAPEIMLNWMHYNQTVDIWSVGCIMAELLTGRTLFPGTDHIDQLKLILRLVG
TPGAELLKKISSESARNYIQSLAQMPKMNFANVFIGANPLAVDLLEKMLVLDSDKRITAA
QALAHAYFAQYHDPDDEPVADPYDQSFESRDLLIDEWKSLTYDEVISFVPPPLDQEEMES
|
|
|
|---|
| BDBM50122915 |
|---|
| n/a |
|---|
| Name | BDBM50122915 |
|---|
| Synonyms: | 2-(2,6-Dichloro-phenyl)-6-[2-(2,4-difluoro-phenyl)-imidazo[1,2-a]pyridin-3-yl]-4,5-dihydro-2H-pyridazin-3-one | 2-(2,6-dichlorophenyl)-6-(2-(2,4-difluorophenyl)imidazo[1,2-a]pyridin-3-yl)-4,5-dihydropyridazin-3(2H)-one | CHEMBL334215 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C23H14Cl2F2N4O |
|---|
| Mol. Mass. | 471.286 |
|---|
| SMILES | Fc1ccc(-c2nc3ccccn3c2C2=NN(C(=O)CC2)c2c(Cl)cccc2Cl)c(F)c1 |t:16| |
|---|
| Structure | 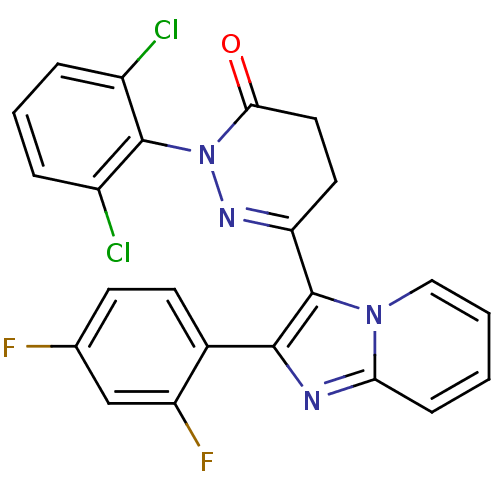
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Colletti, SL; Frie, JL; Dixon, EC; Singh, SB; Choi, BK; Scapin, G; Fitzgerald, CE; Kumar, S; Nichols, EA; O'Keefe, SJ; O'Neill, EA; Porter, G; Samuel, K; Schmatz, DM; Schwartz, CD; Shoop, WL; Thompson, CM; Thompson, JE; Wang, R; Woods, A; Zaller, DM; Doherty, JB Hybrid-designed inhibitors of p38 MAP kinase utilizing N-arylpyridazinones. J Med Chem46:349-52 (2003) [PubMed] Article
Colletti, SL; Frie, JL; Dixon, EC; Singh, SB; Choi, BK; Scapin, G; Fitzgerald, CE; Kumar, S; Nichols, EA; O'Keefe, SJ; O'Neill, EA; Porter, G; Samuel, K; Schmatz, DM; Schwartz, CD; Shoop, WL; Thompson, CM; Thompson, JE; Wang, R; Woods, A; Zaller, DM; Doherty, JB Hybrid-designed inhibitors of p38 MAP kinase utilizing N-arylpyridazinones. J Med Chem46:349-52 (2003) [PubMed] Article