| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Coagulation factor X |
|---|
| Ligand | BDBM12596 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_48662 (CHEMBL657277) |
|---|
| Ki | 1.1±n/a nM |
|---|
| Citation |  Choi-Sledeski, YM; Kearney, R; Poli, G; Pauls, H; Gardner, C; Gong, Y; Becker, M; Davis, R; Spada, A; Liang, G; Chu, V; Brown, K; Collussi, D; Leadley, R; Rebello, S; Moxey, P; Morgan, S; Bentley, R; Kasiewski, C; Maignan, S; Guilloteau, JP; Mikol, V Discovery of an orally efficacious inhibitor of coagulation factor Xa which incorporates a neutral P1 ligand. J Med Chem46:681-4 (2003) [PubMed] Article Choi-Sledeski, YM; Kearney, R; Poli, G; Pauls, H; Gardner, C; Gong, Y; Becker, M; Davis, R; Spada, A; Liang, G; Chu, V; Brown, K; Collussi, D; Leadley, R; Rebello, S; Moxey, P; Morgan, S; Bentley, R; Kasiewski, C; Maignan, S; Guilloteau, JP; Mikol, V Discovery of an orally efficacious inhibitor of coagulation factor Xa which incorporates a neutral P1 ligand. J Med Chem46:681-4 (2003) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Coagulation factor X |
|---|
| Name: | Coagulation factor X |
|---|
| Synonyms: | Activated coagulation factor X (FXa) | Activated factor Xa heavy chain | Coagulation factor X precursor | Coagulation factor Xa | F10 | FA10_HUMAN | Factor X heavy chain | Factor X light chain | Factor Xa | Stuart factor | Stuart-Prower factor |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 54726.60 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | n/a |
|---|
| Residue: | 488 |
|---|
| Sequence: | MGRPLHLVLLSASLAGLLLLGESLFIRREQANNILARVTRANSFLEEMKKGHLERECMEE
TCSYEEAREVFEDSDKTNEFWNKYKDGDQCETSPCQNQGKCKDGLGEYTCTCLEGFEGKN
CELFTRKLCSLDNGDCDQFCHEEQNSVVCSCARGYTLADNGKACIPTGPYPCGKQTLERR
KRSVAQATSSSGEAPDSITWKPYDAADLDPTENPFDLLDFNQTQPERGDNNLTRIVGGQE
CKDGECPWQALLINEENEGFCGGTILSEFYILTAAHCLYQAKRFKVRVGDRNTEQEEGGE
AVHEVEVVIKHNRFTKETYDFDIAVLRLKTPITFRMNVAPACLPERDWAESTLMTQKTGI
VSGFGRTHEKGRQSTRLKMLEVPYVDRNSCKLSSSFIITQNMFCAGYDTKQEDACQGDSG
GPHVTRFKDTYFVTGIVSWGEGCARKGKYGIYTKVTAFLKWIDRSMKTRGLPKAKSHAPE
VITSSPLK
|
|
|
|---|
| BDBM12596 |
|---|
| n/a |
|---|
| Name | BDBM12596 |
|---|
| Synonyms: | 4-{[(E)-2-(5-CHLOROTHIEN-2-YL)VINYL]SULFONYL}-1-(1H-PYRROLO[3,2-C]PYRIDIN-2-YLMETHYL)PIPERAZIN-2-ONE | 4-{[(E)-2-(5-chlorothiophen-2-yl)ethene]sulfonyl}-1-{1H-pyrrolo[3,2-c]pyridin-2-ylmethyl}piperazin-2-one | CHEMBL423417 | Ketopiperazine | RPR209685 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C18H17ClN4O3S2 |
|---|
| Mol. Mass. | 436.936 |
|---|
| SMILES | Clc1ccc(C=CS(=O)(=O)N2CCN(Cc3cc4cnccc4[nH]3)C(=O)C2)s1 |w:5.4| |
|---|
| Structure | 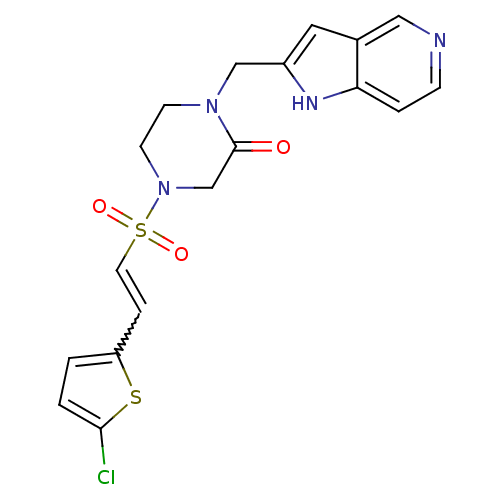
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Choi-Sledeski, YM; Kearney, R; Poli, G; Pauls, H; Gardner, C; Gong, Y; Becker, M; Davis, R; Spada, A; Liang, G; Chu, V; Brown, K; Collussi, D; Leadley, R; Rebello, S; Moxey, P; Morgan, S; Bentley, R; Kasiewski, C; Maignan, S; Guilloteau, JP; Mikol, V Discovery of an orally efficacious inhibitor of coagulation factor Xa which incorporates a neutral P1 ligand. J Med Chem46:681-4 (2003) [PubMed] Article
Choi-Sledeski, YM; Kearney, R; Poli, G; Pauls, H; Gardner, C; Gong, Y; Becker, M; Davis, R; Spada, A; Liang, G; Chu, V; Brown, K; Collussi, D; Leadley, R; Rebello, S; Moxey, P; Morgan, S; Bentley, R; Kasiewski, C; Maignan, S; Guilloteau, JP; Mikol, V Discovery of an orally efficacious inhibitor of coagulation factor Xa which incorporates a neutral P1 ligand. J Med Chem46:681-4 (2003) [PubMed] Article