| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Retinoic acid receptor beta |
|---|
| Ligand | BDBM50032671 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_195644 (CHEMBL795962) |
|---|
| Ki | >1000±n/a nM |
|---|
| Citation |  Michellys, PY; Ardecky, RJ; Chen, JH; D'Arrigo, J; Grese, TA; Karanewsky, DS; Leibowitz, MD; Liu, S; Mais, DA; Mapes, CM; Montrose-Rafizadeh, C; Ogilvie, KM; Reifel-Miller, A; Rungta, D; Thompson, AW; Tyhonas, JS; Boehm, MF Design, synthesis, and structure-activity relationship studies of novel 6,7-locked-[7-(2-alkoxy-3,5-dialkylbenzene)-3-methylocta]-2,4,6-trienoic acids. J Med Chem46:4087-103 (2003) [PubMed] Article Michellys, PY; Ardecky, RJ; Chen, JH; D'Arrigo, J; Grese, TA; Karanewsky, DS; Leibowitz, MD; Liu, S; Mais, DA; Mapes, CM; Montrose-Rafizadeh, C; Ogilvie, KM; Reifel-Miller, A; Rungta, D; Thompson, AW; Tyhonas, JS; Boehm, MF Design, synthesis, and structure-activity relationship studies of novel 6,7-locked-[7-(2-alkoxy-3,5-dialkylbenzene)-3-methylocta]-2,4,6-trienoic acids. J Med Chem46:4087-103 (2003) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Retinoic acid receptor beta |
|---|
| Name: | Retinoic acid receptor beta |
|---|
| Synonyms: | HAP | HBV-activated protein | NR1B2 | Nuclear receptor subfamily 1 group B member 2 | RAR-beta | RAR-epsilon | RARB | RARB_HUMAN | Retinoic acid receptor RXR-alpha/Retinoic acid receptor beta | Retinoic acid receptor beta | Retinoid receptor |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 50498.70 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | ChEMBL_1458015 |
|---|
| Residue: | 455 |
|---|
| Sequence: | MTTSGHACPVPAVNGHMTHYPATPYPLLFPPVIGGLSLPPLHGLHGHPPPSGCSTPSPAT
IETQSTSSEELVPSPPSPLPPPRVYKPCFVCQDKSSGYHYGVSACEGCKGFFRRSIQKNM
IYTCHRDKNCVINKVTRNRCQYCRLQKCFEVGMSKESVRNDRNKKKKETSKQECTESYEM
TAELDDLTEKIRKAHQETFPSLCQLGKYTTNSSADHRVRLDLGLWDKFSELATKCIIKIV
EFAKRLPGFTGLTIADQITLLKAACLDILILRICTRYTPEQDTMTFSDGLTLNRTQMHNA
GFGPLTDLVFTFANQLLPLEMDDTETGLLSAICLICGDRQDLEEPTKVDKLQEPLLEALK
IYIRKRRPSKPHMFPKILMKITDLRSISAKGAERVITLKMEIPGSMPPLIQEMLENSEGH
EPLTPSSSGNTAEHSPSISPSSVENSGVSQSPLVQ
|
|
|
|---|
| BDBM50032671 |
|---|
| n/a |
|---|
| Name | BDBM50032671 |
|---|
| Synonyms: | 6-[1-(3,5,5,8,8-PENTAMETHYL-5,6,7,8-TETRAHYDRONAPHTHALEN-2-YL)CYCLOPROPYL]PYRIDINE-3-CARBOXYLIC ACID | 6-[1-(3,5,5,8,8-Pentamethyl-5,6,7,8-tetrahydro-naphthalen-2-yl)-cyclopropyl]-nicotinic acid | CHEMBL288436 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C24H29NO2 |
|---|
| Mol. Mass. | 363.4926 |
|---|
| SMILES | Cc1cc2c(cc1C1(CC1)c1ccc(cn1)C(O)=O)C(C)(C)CCC2(C)C |
|---|
| Structure | 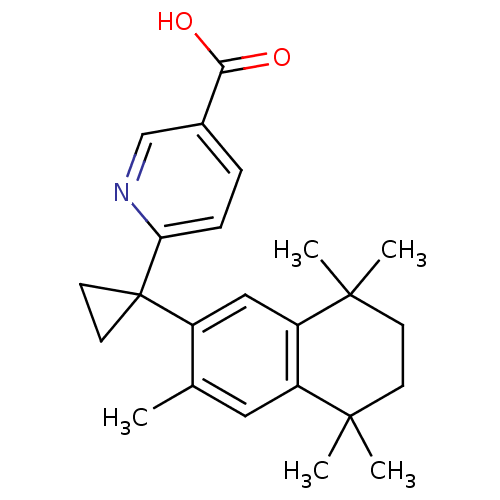
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Michellys, PY; Ardecky, RJ; Chen, JH; D'Arrigo, J; Grese, TA; Karanewsky, DS; Leibowitz, MD; Liu, S; Mais, DA; Mapes, CM; Montrose-Rafizadeh, C; Ogilvie, KM; Reifel-Miller, A; Rungta, D; Thompson, AW; Tyhonas, JS; Boehm, MF Design, synthesis, and structure-activity relationship studies of novel 6,7-locked-[7-(2-alkoxy-3,5-dialkylbenzene)-3-methylocta]-2,4,6-trienoic acids. J Med Chem46:4087-103 (2003) [PubMed] Article
Michellys, PY; Ardecky, RJ; Chen, JH; D'Arrigo, J; Grese, TA; Karanewsky, DS; Leibowitz, MD; Liu, S; Mais, DA; Mapes, CM; Montrose-Rafizadeh, C; Ogilvie, KM; Reifel-Miller, A; Rungta, D; Thompson, AW; Tyhonas, JS; Boehm, MF Design, synthesis, and structure-activity relationship studies of novel 6,7-locked-[7-(2-alkoxy-3,5-dialkylbenzene)-3-methylocta]-2,4,6-trienoic acids. J Med Chem46:4087-103 (2003) [PubMed] Article