| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Prothrombin |
|---|
| Ligand | BDBM50133529 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEBML_208918 |
|---|
| Ki | 0.046000±n/a nM |
|---|
| Citation |  Rittle, KE; Barrow, JC; Cutrona, KJ; Glass, KL; Krueger, JA; Kuo, LC; Lewis, SD; Lucas, BJ; McMasters, DR; Morrissette, MM; Nantermet, PG; Newton, CL; Sanders, WM; Yan, Y; Vacca, JP; Selnick, HG Unexpected enhancement of thrombin inhibitor potency with o-aminoalkylbenzylamides in the P1 position. Bioorg Med Chem Lett13:3477-82 (2003) [PubMed] Rittle, KE; Barrow, JC; Cutrona, KJ; Glass, KL; Krueger, JA; Kuo, LC; Lewis, SD; Lucas, BJ; McMasters, DR; Morrissette, MM; Nantermet, PG; Newton, CL; Sanders, WM; Yan, Y; Vacca, JP; Selnick, HG Unexpected enhancement of thrombin inhibitor potency with o-aminoalkylbenzylamides in the P1 position. Bioorg Med Chem Lett13:3477-82 (2003) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Prothrombin |
|---|
| Name: | Prothrombin |
|---|
| Synonyms: | Activation peptide fragment 1 | Activation peptide fragment 2 | Coagulation factor II | F2 | Prothrombin precursor | THRB_HUMAN | Thrombin heavy chain | Thrombin light chain |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 70029.57 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P00734 |
|---|
| Residue: | 622 |
|---|
| Sequence: | MAHVRGLQLPGCLALAALCSLVHSQHVFLAPQQARSLLQRVRRANTFLEEVRKGNLEREC
VEETCSYEEAFEALESSTATDVFWAKYTACETARTPRDKLAACLEGNCAEGLGTNYRGHV
NITRSGIECQLWRSRYPHKPEINSTTHPGADLQENFCRNPDSSTTGPWCYTTDPTVRRQE
CSIPVCGQDQVTVAMTPRSEGSSVNLSPPLEQCVPDRGQQYQGRLAVTTHGLPCLAWASA
QAKALSKHQDFNSAVQLVENFCRNPDGDEEGVWCYVAGKPGDFGYCDLNYCEEAVEEETG
DGLDEDSDRAIEGRTATSEYQTFFNPRTFGSGEADCGLRPLFEKKSLEDKTERELLESYI
DGRIVEGSDAEIGMSPWQVMLFRKSPQELLCGASLISDRWVLTAAHCLLYPPWDKNFTEN
DLLVRIGKHSRTRYERNIEKISMLEKIYIHPRYNWRENLDRDIALMKLKKPVAFSDYIHP
VCLPDRETAASLLQAGYKGRVTGWGNLKETWTANVGKGQPSVLQVVNLPIVERPVCKDST
RIRITDNMFCAGYKPDEGKRGDACEGDSGGPFVMKSPFNNRWYQMGIVSWGEGCDRDGKY
GFYTHVFRLKKWIQKVIDQFGE
|
|
|
|---|
| BDBM50133529 |
|---|
| n/a |
|---|
| Name | BDBM50133529 |
|---|
| Synonyms: | CHEMBL420682 | N-(2-Aminomethyl-5-chloro-benzyl)-2-(6-methyl-2-oxo-3-phenylmethanesulfonylamino-2H-pyridin-1-yl)-acetamide |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C23H25ClN4O4S |
|---|
| Mol. Mass. | 488.987 |
|---|
| SMILES | Cc1ccc(NS(=O)(=O)Cc2ccccc2)c(=O)n1CC(=O)NCc1cc(Cl)ccc1CN |
|---|
| Structure | 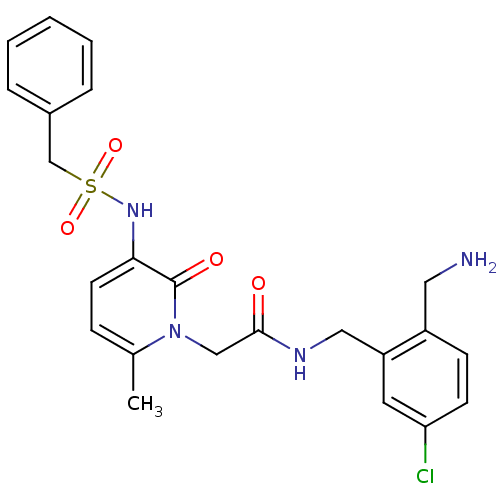
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Rittle, KE; Barrow, JC; Cutrona, KJ; Glass, KL; Krueger, JA; Kuo, LC; Lewis, SD; Lucas, BJ; McMasters, DR; Morrissette, MM; Nantermet, PG; Newton, CL; Sanders, WM; Yan, Y; Vacca, JP; Selnick, HG Unexpected enhancement of thrombin inhibitor potency with o-aminoalkylbenzylamides in the P1 position. Bioorg Med Chem Lett13:3477-82 (2003) [PubMed]
Rittle, KE; Barrow, JC; Cutrona, KJ; Glass, KL; Krueger, JA; Kuo, LC; Lewis, SD; Lucas, BJ; McMasters, DR; Morrissette, MM; Nantermet, PG; Newton, CL; Sanders, WM; Yan, Y; Vacca, JP; Selnick, HG Unexpected enhancement of thrombin inhibitor potency with o-aminoalkylbenzylamides in the P1 position. Bioorg Med Chem Lett13:3477-82 (2003) [PubMed]