| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Reverse transcriptase/RNaseH |
|---|
| Ligand | BDBM50137098 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_196363 (CHEMBL805996) |
|---|
| IC50 | 15100±n/a nM |
|---|
| Citation |  Behforouz, M; Cai, W; Stocksdale, MG; Lucas, JS; Jung, JY; Briere, D; Wang, A; Katen, KS; Behforouz, NC Novel lavendamycin analogues as potent HIV-reverse transcriptase inhibitors: synthesis and evaluation of anti-reverse transcriptase activity of amide and ester analogues of lavendamycin. J Med Chem46:5773-80 (2003) [PubMed] Article Behforouz, M; Cai, W; Stocksdale, MG; Lucas, JS; Jung, JY; Briere, D; Wang, A; Katen, KS; Behforouz, NC Novel lavendamycin analogues as potent HIV-reverse transcriptase inhibitors: synthesis and evaluation of anti-reverse transcriptase activity of amide and ester analogues of lavendamycin. J Med Chem46:5773-80 (2003) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Reverse transcriptase/RNaseH |
|---|
| Name: | Reverse transcriptase/RNaseH |
|---|
| Synonyms: | HIV-1 Reverse Transcriptase RNase H | Human immunodeficiency virus type 1 reverse transcriptase | Reverse transcriptase/RNaseH |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 65229.15 |
|---|
| Organism: | Human immunodeficiency virus 1 |
|---|
| Description: | ChEMBL_1473730 |
|---|
| Residue: | 566 |
|---|
| Sequence: | PISPIETVPVKLKPGMDGPKVKQWPLTEEKIKALVEICTEMEKEGKISKIGPENPYNTPV
FAIKKKDSTKWRKLVDFRELNKRTQDFWEVQLGIPHPAGLKKRKSVTVLDVGDAYFSVPL
DEDFRKYTAFTIPSINNETPGIRYQYNVLPQGWKGSPAIFQSSMTKILEPFRKQNPDIVI
YQYMDDLYVGSDLEIGQHRTKIEELRQHLLRWGLTTPDKKHQKEPPFLWMGYELHPDKWT
VQPIVLPEKDSWTVNDIQKLVGKLNWASQIYPGIRVRQLCKLLRGTKALTEVIPLTEEAE
LELAENREILKEPVHGVYYDPSKDLIAEIQKQGQGQWTYQIYQEPFKNLRTGKYARMRGA
HTNDVKQLTEAVQKITTESIVIWGKTPKFKLPIQKETWETWWTEYWQATWIPEWEFVNTP
PLVKLWYQLEKEPIVGAETFYVDGAANRETKLGKAGYVTNRGRQKVVTLTDTTNQKTELQ
AIYLALQDSGLEVNIVTDSQYALGIIQAQPDQSESELVNQIIEQLIKKEKVYLAWVPAHK
GIGGNEQVDKLVSAGIRKVLFLDGID
|
|
|
|---|
| BDBM50137098 |
|---|
| n/a |
|---|
| Name | BDBM50137098 |
|---|
| Synonyms: | 1-(7-Acetylamino-5,8-dioxo-5,8-dihydro-quinolin-2-yl)-4-methyl-9H-beta-carboline-3-carboxylic acid methyl ester | CHEMBL347211 | methyl 1-(7-acetamido-5,8-dioxo-5,8-dihydroquinolin-2-yl)-4-methyl-9H-pyrido[3,4-b]indole-3-carboxylate |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C25H18N4O5 |
|---|
| Mol. Mass. | 454.4342 |
|---|
| SMILES | COC(=O)c1[nH]c(-c2ccc3C(=O)C=C(NC(C)=O)C(=O)c3n2)c2nc3ccccc3c2c1C |t:13| |
|---|
| Structure | 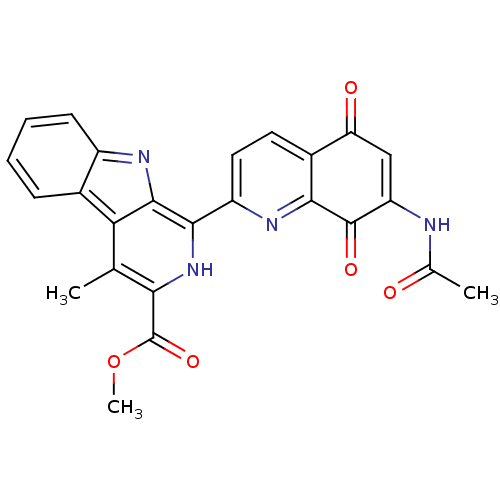
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Behforouz, M; Cai, W; Stocksdale, MG; Lucas, JS; Jung, JY; Briere, D; Wang, A; Katen, KS; Behforouz, NC Novel lavendamycin analogues as potent HIV-reverse transcriptase inhibitors: synthesis and evaluation of anti-reverse transcriptase activity of amide and ester analogues of lavendamycin. J Med Chem46:5773-80 (2003) [PubMed] Article
Behforouz, M; Cai, W; Stocksdale, MG; Lucas, JS; Jung, JY; Briere, D; Wang, A; Katen, KS; Behforouz, NC Novel lavendamycin analogues as potent HIV-reverse transcriptase inhibitors: synthesis and evaluation of anti-reverse transcriptase activity of amide and ester analogues of lavendamycin. J Med Chem46:5773-80 (2003) [PubMed] Article