| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Melanocortin receptor 4 |
|---|
| Ligand | BDBM50139027 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_106179 (CHEMBL714598) |
|---|
| Ki | 34±n/a nM |
|---|
| Citation |  Richardson, TI; Ornstein, PL; Briner, K; Fisher, MJ; Backer, RT; Biggers, CK; Clay, MP; Emmerson, PJ; Hertel, LW; Hsiung, HM; Husain, S; Kahl, SD; Lee, JA; Lindstrom, TD; Martinelli, MJ; Mayer, JP; Mullaney, JT; O'Brien, TP; Pawlak, JM; Revell, KD; Shah, J; Zgombick, JM; Herr, RJ; Melekhov, A; Sampson, PB; King, CH Synthesis and structure-activity relationships of novel arylpiperazines as potent and selective agonists of the melanocortin subtype-4 receptor. J Med Chem47:744-55 (2004) [PubMed] Article Richardson, TI; Ornstein, PL; Briner, K; Fisher, MJ; Backer, RT; Biggers, CK; Clay, MP; Emmerson, PJ; Hertel, LW; Hsiung, HM; Husain, S; Kahl, SD; Lee, JA; Lindstrom, TD; Martinelli, MJ; Mayer, JP; Mullaney, JT; O'Brien, TP; Pawlak, JM; Revell, KD; Shah, J; Zgombick, JM; Herr, RJ; Melekhov, A; Sampson, PB; King, CH Synthesis and structure-activity relationships of novel arylpiperazines as potent and selective agonists of the melanocortin subtype-4 receptor. J Med Chem47:744-55 (2004) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Melanocortin receptor 4 |
|---|
| Name: | Melanocortin receptor 4 |
|---|
| Synonyms: | MC4-R | MC4R | MC4R_HUMAN | Melanocortin MC4 | Melanocortin receptor 4 (MC-4) | Melanocortin receptor 4 (MC4-R) | Melanocortin receptor 4 (MC4R) |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 36949.50 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P32245 |
|---|
| Residue: | 332 |
|---|
| Sequence: | MVNSTHRGMHTSLHLWNRSSYRLHSNASESLGKGYSDGGCYEQLFVSPEVFVTLGVISLL
ENILVIVAIAKNKNLHSPMYFFICSLAVADMLVSVSNGSETIVITLLNSTDTDAQSFTVN
IDNVIDSVICSSLLASICSLLSIAVDRYFTIFYALQYHNIMTVKRVGIIISCIWAACTVS
GILFIIYSDSSAVIICLITMFFTMLALMASLYVHMFLMARLHIKRIAVLPGTGAIRQGAN
MKGAITLTILIGVFVVCWAPFFLHLIFYISCPQNPYCVCFMSHFNLYLILIMCNSIIDPL
IYALRSQELRKTFKEIICCYPLGGLCDLSSRY
|
|
|
|---|
| BDBM50139027 |
|---|
| n/a |
|---|
| Name | BDBM50139027 |
|---|
| Synonyms: | (R)-1,2,3,4-Tetrahydro-isoquinoline-3-carboxylic acid {(R)-1-(4-chloro-benzyl)-2-[4-(2-diethylaminomethyl-phenyl)-piperazin-1-yl]-2-oxo-ethyl}-amide | 1,2,3,4-Tetrahydro-isoquinoline-3-carboxylic acid {(R)-1-(4-chloro-benzyl)-2-[4-(2-diethylaminomethyl-phenyl)-piperazin-1-yl]-2-oxo-ethyl}-amide | CHEMBL433710 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C34H42ClN5O2 |
|---|
| Mol. Mass. | 588.183 |
|---|
| SMILES | CCN(CC)Cc1ccccc1N1CCN(CC1)C(=O)[C@@H](Cc1ccc(Cl)cc1)NC(=O)[C@H]1Cc2ccccc2CN1 |
|---|
| Structure | 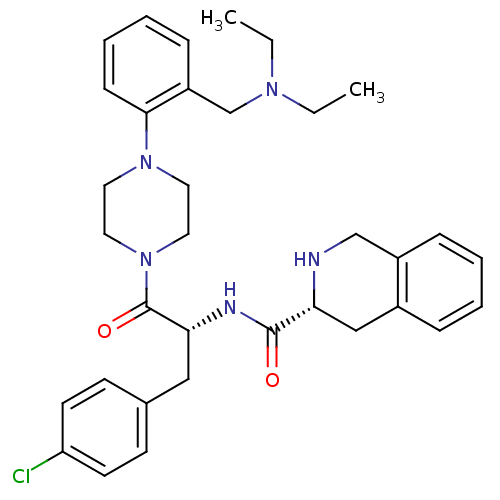
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Richardson, TI; Ornstein, PL; Briner, K; Fisher, MJ; Backer, RT; Biggers, CK; Clay, MP; Emmerson, PJ; Hertel, LW; Hsiung, HM; Husain, S; Kahl, SD; Lee, JA; Lindstrom, TD; Martinelli, MJ; Mayer, JP; Mullaney, JT; O'Brien, TP; Pawlak, JM; Revell, KD; Shah, J; Zgombick, JM; Herr, RJ; Melekhov, A; Sampson, PB; King, CH Synthesis and structure-activity relationships of novel arylpiperazines as potent and selective agonists of the melanocortin subtype-4 receptor. J Med Chem47:744-55 (2004) [PubMed] Article
Richardson, TI; Ornstein, PL; Briner, K; Fisher, MJ; Backer, RT; Biggers, CK; Clay, MP; Emmerson, PJ; Hertel, LW; Hsiung, HM; Husain, S; Kahl, SD; Lee, JA; Lindstrom, TD; Martinelli, MJ; Mayer, JP; Mullaney, JT; O'Brien, TP; Pawlak, JM; Revell, KD; Shah, J; Zgombick, JM; Herr, RJ; Melekhov, A; Sampson, PB; King, CH Synthesis and structure-activity relationships of novel arylpiperazines as potent and selective agonists of the melanocortin subtype-4 receptor. J Med Chem47:744-55 (2004) [PubMed] Article