| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Tissue-type plasminogen activator |
|---|
| Ligand | BDBM50142161 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEBML_208053 |
|---|
| IC50 | >11000±n/a nM |
|---|
| Citation |  Zhang, P; Bao, L; Zuckett, JF; Jia, ZJ; Woolfrey, J; Arfsten, A; Edwards, S; Sinha, U; Hutchaleelaha, A; Lambing, JL; Hollenbach, SJ; Scarborough, RM; Zhu, BY Design, synthesis, and SAR of anthranilamide-based factor Xa inhibitors with improved functional activity. Bioorg Med Chem Lett14:989-93 (2004) [PubMed] Article Zhang, P; Bao, L; Zuckett, JF; Jia, ZJ; Woolfrey, J; Arfsten, A; Edwards, S; Sinha, U; Hutchaleelaha, A; Lambing, JL; Hollenbach, SJ; Scarborough, RM; Zhu, BY Design, synthesis, and SAR of anthranilamide-based factor Xa inhibitors with improved functional activity. Bioorg Med Chem Lett14:989-93 (2004) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Tissue-type plasminogen activator |
|---|
| Name: | Tissue-type plasminogen activator |
|---|
| Synonyms: | Alteplase | PLAT | Reteplase | TPA_HUMAN | Thrombin receptor protein | Tissue-type plasminogen activator | Tissue-type plasminogen activator (tPA) | Tissue-type plasminogen activator precursor | t-PA | t-Plasminogen Activator (tPA) | t-plasminogen activator |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 62931.08 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | n/a |
|---|
| Residue: | 562 |
|---|
| Sequence: | MDAMKRGLCCVLLLCGAVFVSPSQEIHARFRRGARSYQVICRDEKTQMIYQQHQSWLRPV
LRSNRVEYCWCNSGRAQCHSVPVKSCSEPRCFNGGTCQQALYFSDFVCQCPEGFAGKCCE
IDTRATCYEDQGISYRGTWSTAESGAECTNWNSSALAQKPYSGRRPDAIRLGLGNHNYCR
NPDRDSKPWCYVFKAGKYSSEFCSTPACSEGNSDCYFGNGSAYRGTHSLTESGASCLPWN
SMILIGKVYTAQNPSAQALGLGKHNYCRNPDGDAKPWCHVLKNRRLTWEYCDVPSCSTCG
LRQYSQPQFRIKGGLFADIASHPWQAAIFAKHRRSPGERFLCGGILISSCWILSAAHCFQ
ERFPPHHLTVILGRTYRVVPGEEEQKFEVEKYIVHKEFDDDTYDNDIALLQLKSDSSRCA
QESSVVRTVCLPPADLQLPDWTECELSGYGKHEALSPFYSERLKEAHVRLYPSSRCTSQH
LLNRTVTDNMLCAGDTRSGGPQANLHDACQGDSGGPLVCLNDGRMTLVGIISWGLGCGQK
DVPGVYTKVTNYLDWIRDNMRP
|
|
|
|---|
| BDBM50142161 |
|---|
| n/a |
|---|
| Name | BDBM50142161 |
|---|
| Synonyms: | 5-Chloro-N-(5-chloro-pyridin-2-yl)-2-(4-{[methyl-(1-methyl-4,5-dihydro-1H-imidazol-2-yl)-amino]-methyl}-benzoylamino)-benzamide | CHEMBL175130 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C25H24Cl2N6O2 |
|---|
| Mol. Mass. | 511.403 |
|---|
| SMILES | CN(Cc1ccc(cc1)C(=O)Nc1ccc(Cl)cc1C(=O)Nc1ccc(Cl)cn1)C1=NCCN1C |t:32| |
|---|
| Structure | 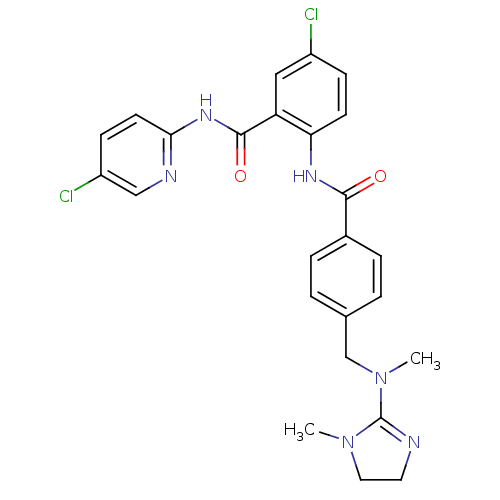
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Zhang, P; Bao, L; Zuckett, JF; Jia, ZJ; Woolfrey, J; Arfsten, A; Edwards, S; Sinha, U; Hutchaleelaha, A; Lambing, JL; Hollenbach, SJ; Scarborough, RM; Zhu, BY Design, synthesis, and SAR of anthranilamide-based factor Xa inhibitors with improved functional activity. Bioorg Med Chem Lett14:989-93 (2004) [PubMed] Article
Zhang, P; Bao, L; Zuckett, JF; Jia, ZJ; Woolfrey, J; Arfsten, A; Edwards, S; Sinha, U; Hutchaleelaha, A; Lambing, JL; Hollenbach, SJ; Scarborough, RM; Zhu, BY Design, synthesis, and SAR of anthranilamide-based factor Xa inhibitors with improved functional activity. Bioorg Med Chem Lett14:989-93 (2004) [PubMed] Article