| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Estrogen receptor |
|---|
| Ligand | BDBM50144847 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_67487 (CHEMBL679324) |
|---|
| IC50 | 1.6±n/a nM |
|---|
| Citation |  Kim, S; Wu, JY; Birzin, ET; Frisch, K; Chan, W; Pai, LY; Yang, YT; Mosley, RT; Fitzgerald, PM; Sharma, N; Dahllund, J; Thorsell, AG; DiNinno, F; Rohrer, SP; Schaeffer, JM; Hammond, ML Estrogen receptor ligands. II. Discovery of benzoxathiins as potent, selective estrogen receptor alpha modulators. J Med Chem47:2171-5 (2004) [PubMed] Article Kim, S; Wu, JY; Birzin, ET; Frisch, K; Chan, W; Pai, LY; Yang, YT; Mosley, RT; Fitzgerald, PM; Sharma, N; Dahllund, J; Thorsell, AG; DiNinno, F; Rohrer, SP; Schaeffer, JM; Hammond, ML Estrogen receptor ligands. II. Discovery of benzoxathiins as potent, selective estrogen receptor alpha modulators. J Med Chem47:2171-5 (2004) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Estrogen receptor |
|---|
| Name: | Estrogen receptor |
|---|
| Synonyms: | ER | ER-alpha | ESR | ESR1 | ESR1_HUMAN | Estradiol receptor | Estrogen receptor | Estrogen receptor (ER alpha) | Estrogen receptor (ER-alpha) | Estrogen receptor alpha (ER alpha) | Estrogen receptor alpha (ER) | NR3A1 | Nuclear receptor subfamily 3 group A member 1 |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 66230.44 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P03372 |
|---|
| Residue: | 595 |
|---|
| Sequence: | MTMTLHTKASGMALLHQIQGNELEPLNRPQLKIPLERPLGEVYLDSSKPAVYNYPEGAAY
EFNAAAAANAQVYGQTGLPYGPGSEAAAFGSNGLGGFPPLNSVSPSPLMLLHPPPQLSPF
LQPHGQQVPYYLENEPSGYTVREAGPPAFYRPNSDNRRQGGRERLASTNDKGSMAMESAK
ETRYCAVCNDYASGYHYGVWSCEGCKAFFKRSIQGHNDYMCPATNQCTIDKNRRKSCQAC
RLRKCYEVGMMKGGIRKDRRGGRMLKHKRQRDDGEGRGEVGSAGDMRAANLWPSPLMIKR
SKKNSLALSLTADQMVSALLDAEPPILYSEYDPTRPFSEASMMGLLTNLADRELVHMINW
AKRVPGFVDLTLHDQVHLLECAWLEILMIGLVWRSMEHPGKLLFAPNLLLDRNQGKCVEG
MVEIFDMLLATSSRFRMMNLQGEEFVCLKSIILLNSGVYTFLSSTLKSLEEKDHIHRVLD
KITDTLIHLMAKAGLTLQQQHQRLAQLLLILSHIRHMSNKGMEHLYSMKCKNVVPLYDLL
LEMLDAHRLHAPTSRGGASVEETDQSHLATAGSTSSHSLQKYYITGEAEGFPATV
|
|
|
|---|
| BDBM50144847 |
|---|
| n/a |
|---|
| Name | BDBM50144847 |
|---|
| Synonyms: | (2R,3S)-3-(4-Hydroxy-phenyl)-2-[4-(2-piperidin-1-yl-ethoxy)-phenyl]-2,3-dihydro-benzo[1,4]oxathiin-7-ol | CHEMBL71584 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C27H29NO4S |
|---|
| Mol. Mass. | 463.588 |
|---|
| SMILES | Oc1ccc(cc1)[C@@H]1Sc2ccc(O)cc2O[C@@H]1c1ccc(OCCN2CCCCC2)cc1 |
|---|
| Structure | 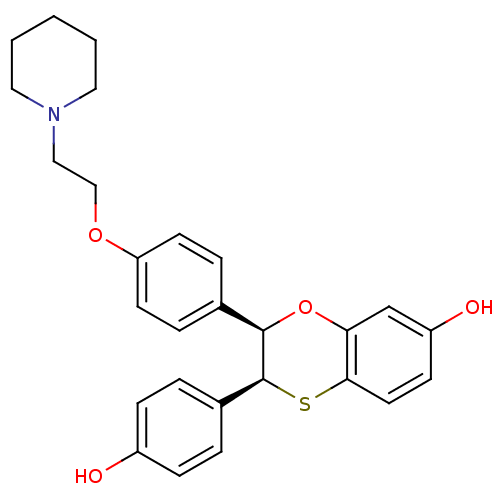
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Kim, S; Wu, JY; Birzin, ET; Frisch, K; Chan, W; Pai, LY; Yang, YT; Mosley, RT; Fitzgerald, PM; Sharma, N; Dahllund, J; Thorsell, AG; DiNinno, F; Rohrer, SP; Schaeffer, JM; Hammond, ML Estrogen receptor ligands. II. Discovery of benzoxathiins as potent, selective estrogen receptor alpha modulators. J Med Chem47:2171-5 (2004) [PubMed] Article
Kim, S; Wu, JY; Birzin, ET; Frisch, K; Chan, W; Pai, LY; Yang, YT; Mosley, RT; Fitzgerald, PM; Sharma, N; Dahllund, J; Thorsell, AG; DiNinno, F; Rohrer, SP; Schaeffer, JM; Hammond, ML Estrogen receptor ligands. II. Discovery of benzoxathiins as potent, selective estrogen receptor alpha modulators. J Med Chem47:2171-5 (2004) [PubMed] Article