| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Alpha-2A adrenergic receptor |
|---|
| Ligand | BDBM50146501 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEBML_33077 |
|---|
| Ki | 0.300000±n/a nM |
|---|
| Citation |  Pastor, J; Alcázar, J; Alvarez, RM; Andrés, JI; Cid, JM; De Lucas, AI; Díaz, A; Fernández, J; Font, LM; Iturrino, L; Lafuente, C; Martínez, S; Bakker, MH; Biesmans, I; Heylen, LI; Megens, AA Synthesis of 3a,4-dihydro-3H-[1]benzopyrano[4,3-c]isoxazoles, displaying combined 5-HT uptake inhibiting and alpha2-adrenoceptor antagonistic activities. Part 2: Further exploration on the cinnamyl moiety. Bioorg Med Chem Lett14:2917-22 (2004) [PubMed] Article Pastor, J; Alcázar, J; Alvarez, RM; Andrés, JI; Cid, JM; De Lucas, AI; Díaz, A; Fernández, J; Font, LM; Iturrino, L; Lafuente, C; Martínez, S; Bakker, MH; Biesmans, I; Heylen, LI; Megens, AA Synthesis of 3a,4-dihydro-3H-[1]benzopyrano[4,3-c]isoxazoles, displaying combined 5-HT uptake inhibiting and alpha2-adrenoceptor antagonistic activities. Part 2: Further exploration on the cinnamyl moiety. Bioorg Med Chem Lett14:2917-22 (2004) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Alpha-2A adrenergic receptor |
|---|
| Name: | Alpha-2A adrenergic receptor |
|---|
| Synonyms: | ADA2A_HUMAN | ADRA2A | ADRA2R | ADRAR | Adrenergic alpha2A | Adrenergic receptor alpha | Alpha-2 adrenergic receptor subtype C10 | Alpha-2A adrenoceptor | Alpha-2A adrenoreceptor | Alpha-2AAR | alpha-2A adrenergic receptor [Homo sapiens] |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 48979.91 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P08913 |
|---|
| Residue: | 465 |
|---|
| Sequence: | MFRQEQPLAEGSFAPMGSLQPDAGNASWNGTEAPGGGARATPYSLQVTLTLVCLAGLLML
LTVFGNVLVIIAVFTSRALKAPQNLFLVSLASADILVATLVIPFSLANEVMGYWYFGKAW
CEIYLALDVLFCTSSIVHLCAISLDRYWSITQAIEYNLKRTPRRIKAIIITVWVISAVIS
FPPLISIEKKGGGGGPQPAEPRCEINDQKWYVISSCIGSFFAPCLIMILVYVRIYQIAKR
RTRVPPSRRGPDAVAAPPGGTERRPNGLGPERSAGPGGAEAEPLPTQLNGAPGEPAPAGP
RDTDALDLEESSSSDHAERPPGPRRPERGPRGKGKARASQVKPGDSLPRRGPGATGIGTP
AAGPGEERVGAAKASRWRGRQNREKRFTFVLAVVIGVFVVCWFPFFFTYTLTAVGCSVPR
TLFKFFFWFGYCNSSLNPVIYTIFNHDFRRAFKKILCRGDRKRIV
|
|
|
|---|
| BDBM50146501 |
|---|
| n/a |
|---|
| Name | BDBM50146501 |
|---|
| Synonyms: | (3R,3aS)-3-{4-[3-(2,5-Difluoro-phenyl)-2-methyl-allyl]-piperazin-1-ylmethyl}-7,8-dimethoxy-3a,4-dihydro-3H-chromeno[4,3-c]isoxazole | CHEMBL319119 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C27H31F2N3O4 |
|---|
| Mol. Mass. | 499.5495 |
|---|
| SMILES | COc1cc2OC[C@H]3[C@H](CN4CCN(C\C(C)=C\c5cc(F)ccc5F)CC4)ON=C3c2cc1OC |c:31| |
|---|
| Structure | 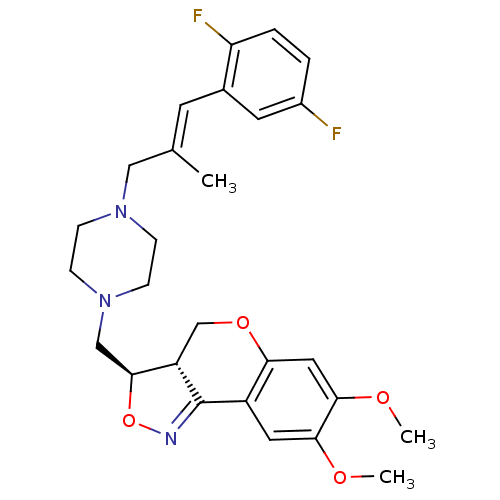
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Pastor, J; Alcázar, J; Alvarez, RM; Andrés, JI; Cid, JM; De Lucas, AI; Díaz, A; Fernández, J; Font, LM; Iturrino, L; Lafuente, C; Martínez, S; Bakker, MH; Biesmans, I; Heylen, LI; Megens, AA Synthesis of 3a,4-dihydro-3H-[1]benzopyrano[4,3-c]isoxazoles, displaying combined 5-HT uptake inhibiting and alpha2-adrenoceptor antagonistic activities. Part 2: Further exploration on the cinnamyl moiety. Bioorg Med Chem Lett14:2917-22 (2004) [PubMed] Article
Pastor, J; Alcázar, J; Alvarez, RM; Andrés, JI; Cid, JM; De Lucas, AI; Díaz, A; Fernández, J; Font, LM; Iturrino, L; Lafuente, C; Martínez, S; Bakker, MH; Biesmans, I; Heylen, LI; Megens, AA Synthesis of 3a,4-dihydro-3H-[1]benzopyrano[4,3-c]isoxazoles, displaying combined 5-HT uptake inhibiting and alpha2-adrenoceptor antagonistic activities. Part 2: Further exploration on the cinnamyl moiety. Bioorg Med Chem Lett14:2917-22 (2004) [PubMed] Article