| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Tissue-type plasminogen activator |
|---|
| Ligand | BDBM50147084 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEBML_208233 |
|---|
| Ki | 870.0±n/a nM |
|---|
| Citation |  Wendt, MD; Geyer, A; McClellan, WJ; Rockway, TW; Weitzberg, M; Zhao, X; Mantei, R; Stewart, K; Nienaber, V; Klinghofer, V; Giranda, VL Interaction with the S1 beta-pocket of urokinase: 8-heterocycle substituted and 6,8-disubstituted 2-naphthamidine urokinase inhibitors. Bioorg Med Chem Lett14:3063-8 (2004) [PubMed] Article Wendt, MD; Geyer, A; McClellan, WJ; Rockway, TW; Weitzberg, M; Zhao, X; Mantei, R; Stewart, K; Nienaber, V; Klinghofer, V; Giranda, VL Interaction with the S1 beta-pocket of urokinase: 8-heterocycle substituted and 6,8-disubstituted 2-naphthamidine urokinase inhibitors. Bioorg Med Chem Lett14:3063-8 (2004) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Tissue-type plasminogen activator |
|---|
| Name: | Tissue-type plasminogen activator |
|---|
| Synonyms: | Alteplase | PLAT | Reteplase | TPA_HUMAN | Thrombin receptor protein | Tissue-type plasminogen activator | Tissue-type plasminogen activator (tPA) | Tissue-type plasminogen activator precursor | t-PA | t-Plasminogen Activator (tPA) | t-plasminogen activator |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 62931.08 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | n/a |
|---|
| Residue: | 562 |
|---|
| Sequence: | MDAMKRGLCCVLLLCGAVFVSPSQEIHARFRRGARSYQVICRDEKTQMIYQQHQSWLRPV
LRSNRVEYCWCNSGRAQCHSVPVKSCSEPRCFNGGTCQQALYFSDFVCQCPEGFAGKCCE
IDTRATCYEDQGISYRGTWSTAESGAECTNWNSSALAQKPYSGRRPDAIRLGLGNHNYCR
NPDRDSKPWCYVFKAGKYSSEFCSTPACSEGNSDCYFGNGSAYRGTHSLTESGASCLPWN
SMILIGKVYTAQNPSAQALGLGKHNYCRNPDGDAKPWCHVLKNRRLTWEYCDVPSCSTCG
LRQYSQPQFRIKGGLFADIASHPWQAAIFAKHRRSPGERFLCGGILISSCWILSAAHCFQ
ERFPPHHLTVILGRTYRVVPGEEEQKFEVEKYIVHKEFDDDTYDNDIALLQLKSDSSRCA
QESSVVRTVCLPPADLQLPDWTECELSGYGKHEALSPFYSERLKEAHVRLYPSSRCTSQH
LLNRTVTDNMLCAGDTRSGGPQANLHDACQGDSGGPLVCLNDGRMTLVGIISWGLGCGQK
DVPGVYTKVTNYLDWIRDNMRP
|
|
|
|---|
| BDBM50147084 |
|---|
| n/a |
|---|
| Name | BDBM50147084 |
|---|
| Synonyms: | 6-Carbamimidoyl-4-(tetrahydro-furan-3-yl)-naphthalene-2-carboxylic acid phenylamide | 6-carbamimidoyl-N-phenyl-4-(tetrahydrofuran-3-yl)-2-naphthamide | CHEMBL105395 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C22H21N3O2 |
|---|
| Mol. Mass. | 359.421 |
|---|
| SMILES | NC(=N)c1ccc2cc(cc(C3CCOC3)c2c1)C(=O)Nc1ccccc1 |
|---|
| Structure | 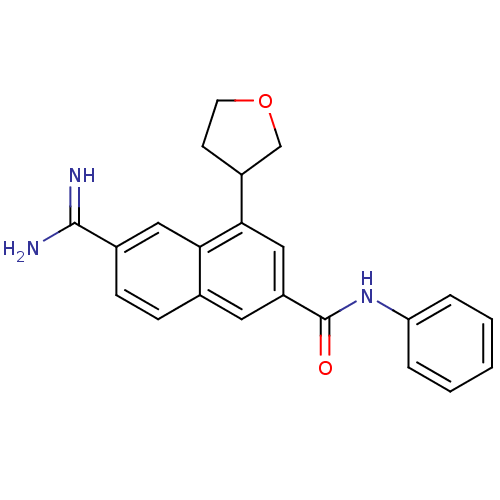
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Wendt, MD; Geyer, A; McClellan, WJ; Rockway, TW; Weitzberg, M; Zhao, X; Mantei, R; Stewart, K; Nienaber, V; Klinghofer, V; Giranda, VL Interaction with the S1 beta-pocket of urokinase: 8-heterocycle substituted and 6,8-disubstituted 2-naphthamidine urokinase inhibitors. Bioorg Med Chem Lett14:3063-8 (2004) [PubMed] Article
Wendt, MD; Geyer, A; McClellan, WJ; Rockway, TW; Weitzberg, M; Zhao, X; Mantei, R; Stewart, K; Nienaber, V; Klinghofer, V; Giranda, VL Interaction with the S1 beta-pocket of urokinase: 8-heterocycle substituted and 6,8-disubstituted 2-naphthamidine urokinase inhibitors. Bioorg Med Chem Lett14:3063-8 (2004) [PubMed] Article