| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Corticotropin-releasing factor receptor 1 |
|---|
| Ligand | BDBM50155964 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_306679 (CHEMBL830013) |
|---|
| IC50 | 0.5±n/a nM |
|---|
| Citation |  Dzierba, CD; Takvorian, AG; Rafalski, M; Kasireddy-Polam, P; Wong, H; Molski, TF; Zhang, G; Li, YW; Lelas, S; Peng, Y; McElroy, JF; Zaczek, RC; Taub, RA; Combs, AP; Gilligan, PJ; Trainor, GL Synthesis, structure-activity relationships, and in vivo properties of 3,4-dihydro-1H-pyrido[2,3-b]pyrazin-2-ones as corticotropin-releasing factor-1 receptor antagonists. J Med Chem47:5783-90 (2004) [PubMed] Article Dzierba, CD; Takvorian, AG; Rafalski, M; Kasireddy-Polam, P; Wong, H; Molski, TF; Zhang, G; Li, YW; Lelas, S; Peng, Y; McElroy, JF; Zaczek, RC; Taub, RA; Combs, AP; Gilligan, PJ; Trainor, GL Synthesis, structure-activity relationships, and in vivo properties of 3,4-dihydro-1H-pyrido[2,3-b]pyrazin-2-ones as corticotropin-releasing factor-1 receptor antagonists. J Med Chem47:5783-90 (2004) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Corticotropin-releasing factor receptor 1 |
|---|
| Name: | Corticotropin-releasing factor receptor 1 |
|---|
| Synonyms: | CRF-R | CRF1 | CRFR1_RAT | CRH-R 1 | Corticotropin releasing factor receptor | Corticotropin releasing factor receptor 1 | Corticotropin-releasing Factor Receptor 1 | Corticotropin-releasing hormone receptor 1 | Crhr | Crhr1 |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 47870.75 |
|---|
| Organism: | Rattus norvegicus (rat) |
|---|
| Description: | Receptor binding assays were performed using rat cortex homogenate. |
|---|
| Residue: | 415 |
|---|
| Sequence: | MGRRPQLRLVKALLLLGLNPVSTSLQDQRCENLSLTSNVSGLQCNASVDLIGTCWPRSPA
GQLVVRPCPAFFYGVRYNTTNNGYRECLANGSWAARVNYSECQEILNEEKKSKVHYHVAV
IINYLGHCISLVALLVAFVLFLRLRSIRCLRNIIHWNLISAFILRNATWFVVQLTVSPEV
HQSNVAWCRLVTAAYNYFHVTNFFWMFGEGCYLHTAIVLTYSTDRLRKWMFVCIGWGVPF
PIIVAWAIGKLHYDNEKCWFGKRPGVYTDYIYQGPMILVLLINFIFLFNIVRILMTKLRA
STTSETIQYRKAVKATLVLLPLLGITYMLFFVNPGEDEVSRVVFIYFNSFLESFQGFFVS
VFYCFLNSEVRSAIRKRWRRWQDKHSIRARVARAMSIPTSPTRVSFHSIKQSTAV
|
|
|
|---|
| BDBM50155964 |
|---|
| n/a |
|---|
| Name | BDBM50155964 |
|---|
| Synonyms: | 8-(Butyl-ethyl-amino)-4-(2-chloro-5-fluoro-4-methoxy-phenyl)-6-methyl-3,4-dihydro-1H-pyrido[2,3-b]pyrazin-2-one; TFA | CHEMBL188464 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C21H26ClFN4O2 |
|---|
| Mol. Mass. | 420.908 |
|---|
| SMILES | CCCCN(CC)c1cc(C)nc2N(CC(=O)Nc12)c1cc(F)c(OC)cc1Cl |
|---|
| Structure | 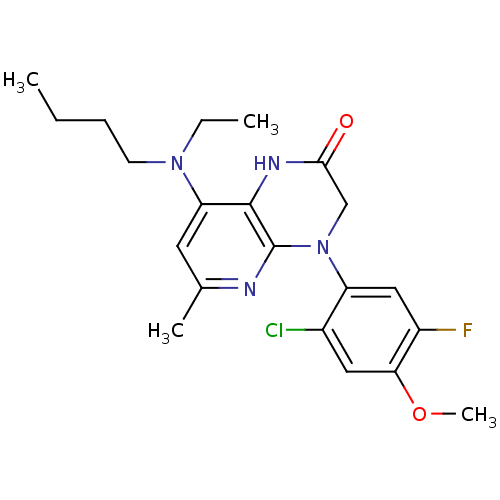
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Dzierba, CD; Takvorian, AG; Rafalski, M; Kasireddy-Polam, P; Wong, H; Molski, TF; Zhang, G; Li, YW; Lelas, S; Peng, Y; McElroy, JF; Zaczek, RC; Taub, RA; Combs, AP; Gilligan, PJ; Trainor, GL Synthesis, structure-activity relationships, and in vivo properties of 3,4-dihydro-1H-pyrido[2,3-b]pyrazin-2-ones as corticotropin-releasing factor-1 receptor antagonists. J Med Chem47:5783-90 (2004) [PubMed] Article
Dzierba, CD; Takvorian, AG; Rafalski, M; Kasireddy-Polam, P; Wong, H; Molski, TF; Zhang, G; Li, YW; Lelas, S; Peng, Y; McElroy, JF; Zaczek, RC; Taub, RA; Combs, AP; Gilligan, PJ; Trainor, GL Synthesis, structure-activity relationships, and in vivo properties of 3,4-dihydro-1H-pyrido[2,3-b]pyrazin-2-ones as corticotropin-releasing factor-1 receptor antagonists. J Med Chem47:5783-90 (2004) [PubMed] Article