| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Histamine H2 receptor |
|---|
| Ligand | BDBM50156866 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_429753 (CHEMBL914453) |
|---|
| IC50 | 3000±n/a nM |
|---|
| Citation |  Fonquerna, S; Miralpeix, M; Pagès, L; Puig, C; Cardús, A; Antón, F; Cárdenas, A; Vilella, D; Aparici, M; Calaf, E; Prieto, J; Gras, J; Huerta, JM; Warrellow, G; Beleta, J; Ryder, H Synthesis and structure-activity relationships of novel histamine H1 antagonists: indolylpiperidinyl benzoic acid derivatives. J Med Chem47:6326-37 (2004) [PubMed] Article Fonquerna, S; Miralpeix, M; Pagès, L; Puig, C; Cardús, A; Antón, F; Cárdenas, A; Vilella, D; Aparici, M; Calaf, E; Prieto, J; Gras, J; Huerta, JM; Warrellow, G; Beleta, J; Ryder, H Synthesis and structure-activity relationships of novel histamine H1 antagonists: indolylpiperidinyl benzoic acid derivatives. J Med Chem47:6326-37 (2004) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Histamine H2 receptor |
|---|
| Name: | Histamine H2 receptor |
|---|
| Synonyms: | Gastric receptor I | H2R | HISTAMINE H2 | HRH2 | HRH2_HUMAN | Histamine H2 receptor | Histamine H2-Gs alpha S |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 40115.31 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | n/a |
|---|
| Residue: | 359 |
|---|
| Sequence: | MAPNGTASSFCLDSTACKITITVVLAVLILITVAGNVVVCLAVGLNRRLRNLTNCFIVSL
AITDLLLGLLVLPFSAIYQLSCKWSFGKVFCNIYTSLDVMLCTASILNLFMISLDRYCAV
MDPLRYPVLVTPVRVAISLVLIWVISITLSFLSIHLGWNSRNETSKGNHTTSKCKVQVNE
VYGLVDGLVTFYLPLLIMCITYYRIFKVARDQAKRINHISSWKAATIREHKATVTLAAVM
GAFIICWFPYFTAFVYRGLRGDDAINEVLEAIVLWLGYANSALNPILYAALNRDFRTGYQ
QLFCCRLANRNSHKTSLRSNASQLSRTQSREPRQQEEKPLKLQVWSGTEVTAPQGATDR
|
|
|
|---|
| BDBM50156866 |
|---|
| n/a |
|---|
| Name | BDBM50156866 |
|---|
| Synonyms: | 5-[4-(6-fluoro-1-thiophen-2-ylmethyl-1H-indol-3-yl)piperidin-1-ylmethyl]-2-methoxybenzoic acid | CHEMBL222139 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C27H27FN2O3S |
|---|
| Mol. Mass. | 478.578 |
|---|
| SMILES | COc1ccc(CN2CCC(CC2)c2cn(Cc3cccs3)c3cc(F)ccc23)cc1C(O)=O |
|---|
| Structure | 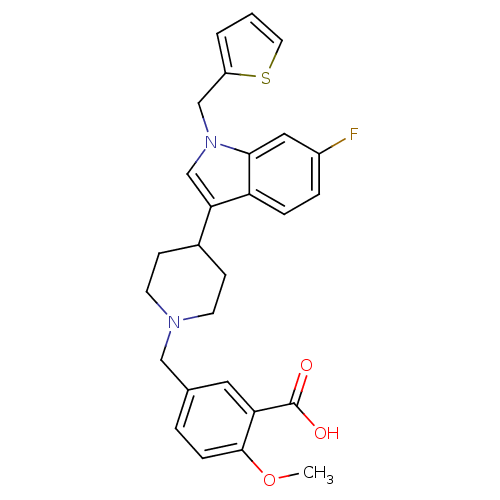
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Fonquerna, S; Miralpeix, M; Pagès, L; Puig, C; Cardús, A; Antón, F; Cárdenas, A; Vilella, D; Aparici, M; Calaf, E; Prieto, J; Gras, J; Huerta, JM; Warrellow, G; Beleta, J; Ryder, H Synthesis and structure-activity relationships of novel histamine H1 antagonists: indolylpiperidinyl benzoic acid derivatives. J Med Chem47:6326-37 (2004) [PubMed] Article
Fonquerna, S; Miralpeix, M; Pagès, L; Puig, C; Cardús, A; Antón, F; Cárdenas, A; Vilella, D; Aparici, M; Calaf, E; Prieto, J; Gras, J; Huerta, JM; Warrellow, G; Beleta, J; Ryder, H Synthesis and structure-activity relationships of novel histamine H1 antagonists: indolylpiperidinyl benzoic acid derivatives. J Med Chem47:6326-37 (2004) [PubMed] Article