| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Retinoic acid receptor alpha |
|---|
| Ligand | BDBM50091698 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_430404 (CHEMBL916388) |
|---|
| IC50 | 20000±n/a nM |
|---|
| Citation |  Patel, JB; Huynh, CK; Handratta, VD; Gediya, LK; Brodie, AM; Goloubeva, OG; Clement, OO; Nanne, IP; Soprano, DR; Njar, VC Novel retinoic acid metabolism blocking agents endowed with multiple biological activities are efficient growth inhibitors of human breast and prostate cancer cells in vitro and a human breast tumor xenograft in nude mice. J Med Chem47:6716-29 (2004) [PubMed] Article Patel, JB; Huynh, CK; Handratta, VD; Gediya, LK; Brodie, AM; Goloubeva, OG; Clement, OO; Nanne, IP; Soprano, DR; Njar, VC Novel retinoic acid metabolism blocking agents endowed with multiple biological activities are efficient growth inhibitors of human breast and prostate cancer cells in vitro and a human breast tumor xenograft in nude mice. J Med Chem47:6716-29 (2004) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Retinoic acid receptor alpha |
|---|
| Name: | Retinoic acid receptor alpha |
|---|
| Synonyms: | NR1B1 | Nuclear receptor subfamily 1 group B member 1 | RAR-alpha | RARA | RARA_HUMAN | Retinoic acid receptor alpha | Retinoic acid receptor alpha/Retinoid X receptor alpha | Retinoid X receptor gamma/retinoic acid receptor alpha | Retinoid receptor |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 50778.87 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | ChEMBL_1466191 |
|---|
| Residue: | 462 |
|---|
| Sequence: | MASNSSSCPTPGGGHLNGYPVPPYAFFFPPMLGGLSPPGALTTLQHQLPVSGYSTPSPAT
IETQSSSSEEIVPSPPSPPPLPRIYKPCFVCQDKSSGYHYGVSACEGCKGFFRRSIQKNM
VYTCHRDKNCIINKVTRNRCQYCRLQKCFEVGMSKESVRNDRNKKKKEVPKPECSESYTL
TPEVGELIEKVRKAHQETFPALCQLGKYTTNNSSEQRVSLDIDLWDKFSELSTKCIIKTV
EFAKQLPGFTTLTIADQITLLKAACLDILILRICTRYTPEQDTMTFSDGLTLNRTQMHNA
GFGPLTDLVFAFANQLLPLEMDDAETGLLSAICLICGDRQDLEQPDRVDMLQEPLLEALK
VYVRKRRPSRPHMFPKMLMKITDLRSISAKGAERVITLKMEIPGSMPPLIQEMLENSEGL
DTLSGQPGGGGRDGGGLAPPPGSCSPSLSPSSNRSSPATHSP
|
|
|
|---|
| BDBM50091698 |
|---|
| n/a |
|---|
| Name | BDBM50091698 |
|---|
| Synonyms: | (2E,4E,6E,8E)-9-(3-Imidazol-1-yl-2,6,6-trimethyl-cyclohex-1-enyl)-3,7-dimethyl-nona-2,4,6,8-tetraenoic acid | 4-((+/-)-(1H-imidazol-1-yl)-(E)-retinoic acid | CHEMBL57765 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C23H30N2O2 |
|---|
| Mol. Mass. | 366.4965 |
|---|
| SMILES | C\C(\C=C\C1=C(C)C(CCC1(C)C)n1ccnc1)=C/C=C/C(/C)=C/C(O)=O |c:4| |
|---|
| Structure | 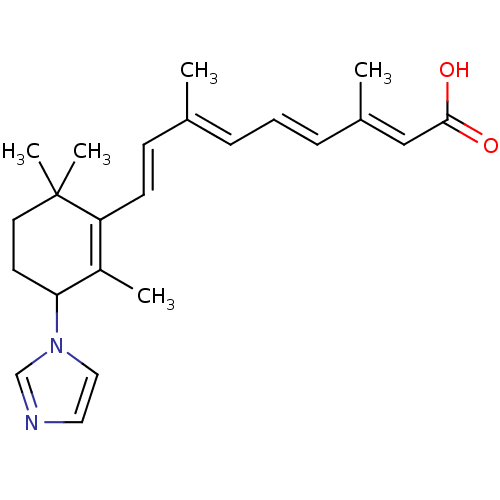
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Patel, JB; Huynh, CK; Handratta, VD; Gediya, LK; Brodie, AM; Goloubeva, OG; Clement, OO; Nanne, IP; Soprano, DR; Njar, VC Novel retinoic acid metabolism blocking agents endowed with multiple biological activities are efficient growth inhibitors of human breast and prostate cancer cells in vitro and a human breast tumor xenograft in nude mice. J Med Chem47:6716-29 (2004) [PubMed] Article
Patel, JB; Huynh, CK; Handratta, VD; Gediya, LK; Brodie, AM; Goloubeva, OG; Clement, OO; Nanne, IP; Soprano, DR; Njar, VC Novel retinoic acid metabolism blocking agents endowed with multiple biological activities are efficient growth inhibitors of human breast and prostate cancer cells in vitro and a human breast tumor xenograft in nude mice. J Med Chem47:6716-29 (2004) [PubMed] Article