| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Mitogen-activated protein kinase 11 |
|---|
| Ligand | BDBM13336 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_325099 (CHEMBL860916) |
|---|
| Kd | 250±n/a nM |
|---|
| Citation |  Fabian, MA; Biggs, WH; Treiber, DK; Atteridge, CE; Azimioara, MD; Benedetti, MG; Carter, TA; Ciceri, P; Edeen, PT; Floyd, M; Ford, JM; Galvin, M; Gerlach, JL; Grotzfeld, RM; Herrgard, S; Insko, DE; Insko, MA; Lai, AG; Lélias, JM; Mehta, SA; Milanov, ZV; Velasco, AM; Wodicka, LM; Patel, HK; Zarrinkar, PP; Lockhart, DJ A small molecule-kinase interaction map for clinical kinase inhibitors. Nat Biotechnol23:329-36 (2005) [PubMed] Article Fabian, MA; Biggs, WH; Treiber, DK; Atteridge, CE; Azimioara, MD; Benedetti, MG; Carter, TA; Ciceri, P; Edeen, PT; Floyd, M; Ford, JM; Galvin, M; Gerlach, JL; Grotzfeld, RM; Herrgard, S; Insko, DE; Insko, MA; Lai, AG; Lélias, JM; Mehta, SA; Milanov, ZV; Velasco, AM; Wodicka, LM; Patel, HK; Zarrinkar, PP; Lockhart, DJ A small molecule-kinase interaction map for clinical kinase inhibitors. Nat Biotechnol23:329-36 (2005) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Mitogen-activated protein kinase 11 |
|---|
| Name: | Mitogen-activated protein kinase 11 |
|---|
| Synonyms: | MAP kinase p38 | MAPK11 | MK11_HUMAN | Mitogen-activated protein kinase 11 | Mitogen-activated protein kinase p38 beta | PRKM11 | SAPK2 | SAPK2B | Stress-activated protein kinase 2 | p38 MAP kinase alpha/beta | p38-2 | p38-beta | p38b |
|---|
| Type: | Enzyme Catalytic Domain |
|---|
| Mol. Mass.: | 41351.73 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | gi_20128774 |
|---|
| Residue: | 364 |
|---|
| Sequence: | MSGPRAGFYRQELNKTVWEVPQRLQGLRPVGSGAYGSVCSAYDARLRQKVAVKKLSRPFQ
SLIHARRTYRELRLLKHLKHENVIGLLDVFTPATSIEDFSEVYLVTTLMGADLNNIVKCQ
ALSDEHVQFLVYQLLRGLKYIHSAGIIHRDLKPSNVAVNEDCELRILDFGLARQADEEMT
GYVATRWYRAPEIMLNWMHYNQTVDIWSVGCIMAELLQGKALFPGSDYIDQLKRIMEVVG
TPSPEVLAKISSEHARTYIQSLPPMPQKDLSSIFRGANPLAIDLLGRMLVLDSDQRVSAA
EALAHAYFSQYHDPEDEPEAEPYDESVEAKERTLEEWKELTYQEVLSFKPPEPPKPPGSL
EIEQ
|
|
|
|---|
| BDBM13336 |
|---|
| n/a |
|---|
| Name | BDBM13336 |
|---|
| Synonyms: | 4-[4-(4-fluorophenyl)-2-(4-methanesulfinylphenyl)-1H-imidazol-5-yl]pyridine | 4-[4-(4-fluorophenyl)-2-(4-methylsulfinylphenyl)-1H-imidazol-5-yl]pyridine | 4-{4-(4-fluorophenyl)-2-[4-(methylsulfinyl)phenyl]-1H-imidazol-5-yl}pyridine | CHEMBL10 | SB-203580 | SB203580 | cid_176155 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C21H16FN3OS |
|---|
| Mol. Mass. | 377.435 |
|---|
| SMILES | CS(=O)c1ccc(cc1)-c1nc(c([nH]1)-c1ccc(F)cc1)-c1ccncc1 |
|---|
| Structure | 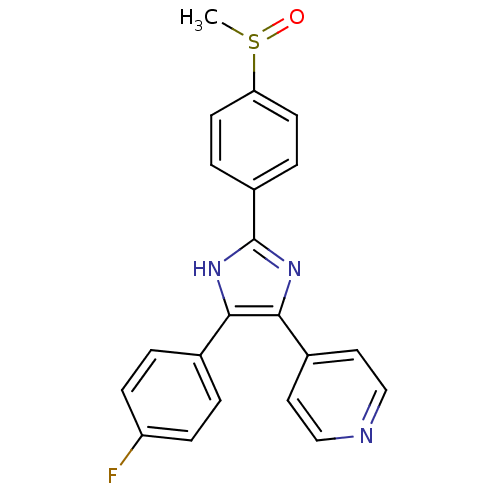
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Fabian, MA; Biggs, WH; Treiber, DK; Atteridge, CE; Azimioara, MD; Benedetti, MG; Carter, TA; Ciceri, P; Edeen, PT; Floyd, M; Ford, JM; Galvin, M; Gerlach, JL; Grotzfeld, RM; Herrgard, S; Insko, DE; Insko, MA; Lai, AG; Lélias, JM; Mehta, SA; Milanov, ZV; Velasco, AM; Wodicka, LM; Patel, HK; Zarrinkar, PP; Lockhart, DJ A small molecule-kinase interaction map for clinical kinase inhibitors. Nat Biotechnol23:329-36 (2005) [PubMed] Article
Fabian, MA; Biggs, WH; Treiber, DK; Atteridge, CE; Azimioara, MD; Benedetti, MG; Carter, TA; Ciceri, P; Edeen, PT; Floyd, M; Ford, JM; Galvin, M; Gerlach, JL; Grotzfeld, RM; Herrgard, S; Insko, DE; Insko, MA; Lai, AG; Lélias, JM; Mehta, SA; Milanov, ZV; Velasco, AM; Wodicka, LM; Patel, HK; Zarrinkar, PP; Lockhart, DJ A small molecule-kinase interaction map for clinical kinase inhibitors. Nat Biotechnol23:329-36 (2005) [PubMed] Article