| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 3A4 |
|---|
| Ligand | BDBM50162783 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_305432 (CHEMBL830925) |
|---|
| IC50 | 4±n/a nM |
|---|
| Citation |  Mulvihill, MJ; Kan, JL; Beck, P; Bittner, M; Cesario, C; Cooke, A; Keane, DM; Nigro, AI; Nillson, C; Smith, V; Srebernak, M; Sun, FL; Vrkljan, M; Winski, SL; Castelhano, AL; Emerson, D; Gibson, N Potent and selective [2-imidazol-1-yl-2-(6-alkoxy-naphthalen-2-yl)-1-methyl-ethyl]-dimethyl-amines as retinoic acid metabolic blocking agents (RAMBAs). Bioorg Med Chem Lett15:1669-73 (2005) [PubMed] Article Mulvihill, MJ; Kan, JL; Beck, P; Bittner, M; Cesario, C; Cooke, A; Keane, DM; Nigro, AI; Nillson, C; Smith, V; Srebernak, M; Sun, FL; Vrkljan, M; Winski, SL; Castelhano, AL; Emerson, D; Gibson, N Potent and selective [2-imidazol-1-yl-2-(6-alkoxy-naphthalen-2-yl)-1-methyl-ethyl]-dimethyl-amines as retinoic acid metabolic blocking agents (RAMBAs). Bioorg Med Chem Lett15:1669-73 (2005) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 3A4 |
|---|
| Name: | Cytochrome P450 3A4 |
|---|
| Synonyms: | Albendazole monooxygenase | Albendazole sulfoxidase | CP3A4_HUMAN | CYP3A3 | CYP3A4 | CYPIIIA3 | CYPIIIA4 | Cytochrome P450 3A3 | Cytochrome P450 3A4 (CYP3A4) | Cytochrome P450 HLp | Nifedipine oxidase | Quinine 3-monooxygenase | Taurochenodeoxycholate 6-alpha-hydroxylase |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 57349.57 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | n/a |
|---|
| Residue: | 503 |
|---|
| Sequence: | MALIPDLAMETWLLLAVSLVLLYLYGTHSHGLFKKLGIPGPTPLPFLGNILSYHKGFCMF
DMECHKKYGKVWGFYDGQQPVLAITDPDMIKTVLVKECYSVFTNRRPFGPVGFMKSAISI
AEDEEWKRLRSLLSPTFTSGKLKEMVPIIAQYGDVLVRNLRREAETGKPVTLKDVFGAYS
MDVITSTSFGVNIDSLNNPQDPFVENTKKLLRFDFLDPFFLSITVFPFLIPILEVLNICV
FPREVTNFLRKSVKRMKESRLEDTQKHRVDFLQLMIDSQNSKETESHKALSDLELVAQSI
IFIFAGYETTSSVLSFIMYELATHPDVQQKLQEEIDAVLPNKAPPTYDTVLQMEYLDMVV
NETLRLFPIAMRLERVCKKDVEINGMFIPKGVVVMIPSYALHRDPKYWTEPEKFLPERFS
KKNKDNIDPYIYTPFGSGPRNCIGMRFALMNMKLALIRVLQNFSFKPCKETQIPLKLSLG
GLLQPEKPVVLKVESRDGTVSGA
|
|
|
|---|
| BDBM50162783 |
|---|
| n/a |
|---|
| Name | BDBM50162783 |
|---|
| Synonyms: | CHEMBL179825 | {4-[6-(2-Dimethylamino-1-imidazol-1-yl-propyl)-naphthalen-2-yloxymethyl]-phenyl}-acetic acid methyl ester |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C28H31N3O3 |
|---|
| Mol. Mass. | 457.564 |
|---|
| SMILES | COC(=O)Cc1ccc(COc2ccc3cc(ccc3c2)C(C(C)N(C)C)n2ccnc2)cc1 |
|---|
| Structure | 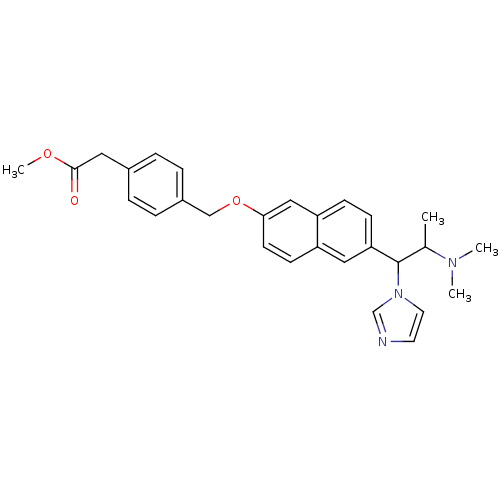
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Mulvihill, MJ; Kan, JL; Beck, P; Bittner, M; Cesario, C; Cooke, A; Keane, DM; Nigro, AI; Nillson, C; Smith, V; Srebernak, M; Sun, FL; Vrkljan, M; Winski, SL; Castelhano, AL; Emerson, D; Gibson, N Potent and selective [2-imidazol-1-yl-2-(6-alkoxy-naphthalen-2-yl)-1-methyl-ethyl]-dimethyl-amines as retinoic acid metabolic blocking agents (RAMBAs). Bioorg Med Chem Lett15:1669-73 (2005) [PubMed] Article
Mulvihill, MJ; Kan, JL; Beck, P; Bittner, M; Cesario, C; Cooke, A; Keane, DM; Nigro, AI; Nillson, C; Smith, V; Srebernak, M; Sun, FL; Vrkljan, M; Winski, SL; Castelhano, AL; Emerson, D; Gibson, N Potent and selective [2-imidazol-1-yl-2-(6-alkoxy-naphthalen-2-yl)-1-methyl-ethyl]-dimethyl-amines as retinoic acid metabolic blocking agents (RAMBAs). Bioorg Med Chem Lett15:1669-73 (2005) [PubMed] Article