| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Basic phospholipase A2 1 |
|---|
| Ligand | BDBM50148911 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_305607 (CHEMBL828145) |
|---|
| IC50 | 2900±n/a nM |
|---|
| Citation |  Khanum, SA; Murari, SK; Vishwanth, BS; Shashikanth, S Synthesis of benzoyl phenyl benzoates as effective inhibitors for phospholipase A2 and hyaluronidase enzymes. Bioorg Med Chem Lett15:4100-4 (2005) [PubMed] Article Khanum, SA; Murari, SK; Vishwanth, BS; Shashikanth, S Synthesis of benzoyl phenyl benzoates as effective inhibitors for phospholipase A2 and hyaluronidase enzymes. Bioorg Med Chem Lett15:4100-4 (2005) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Basic phospholipase A2 1 |
|---|
| Name: | Basic phospholipase A2 1 |
|---|
| Synonyms: | PA2B1_NAJME | Phospholipase A2 isozyme DE-I |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 13475.55 |
|---|
| Organism: | Naja melanoleuca |
|---|
| Description: | ChEMBL_305607 |
|---|
| Residue: | 118 |
|---|
| Sequence: | NLYQFKNMIHCTVPNRPWWHFANYGCYCGRGGKGTPVDDLDRCCQIHDKCYDEAEKISGC
WPYIKTYTYESCQGTLTCKDGGKCAASVCDCDRVAANCFARATYNDKNYNIDFNARCQ
|
|
|
|---|
| BDBM50148911 |
|---|
| n/a |
|---|
| Name | BDBM50148911 |
|---|
| Synonyms: | (3beta)-3-hydroxyurs-12-en-28-oic acid | 3beta-hydroxyurs-12-en-28-oic acid | CHEMBL169 | Ursolic acid | malol | prunol | urson |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C30H48O3 |
|---|
| Mol. Mass. | 456.7003 |
|---|
| SMILES | C[C@@H]1CC[C@@]2(CC[C@]3(C)C(=CC[C@@H]4[C@@]5(C)CC[C@H](O)C(C)(C)[C@@H]5CC[C@@]34C)[C@@H]2[C@H]1C)C(O)=O |r,c:9| |
|---|
| Structure | 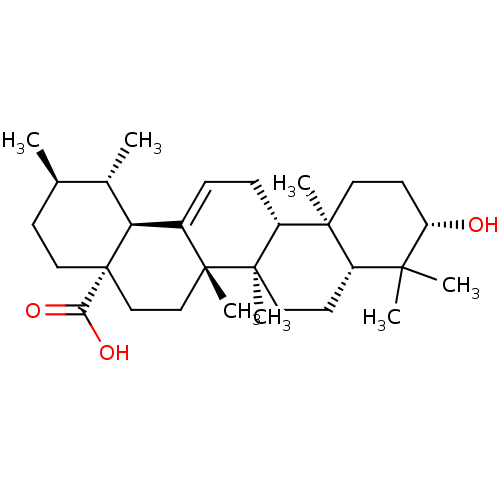
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Khanum, SA; Murari, SK; Vishwanth, BS; Shashikanth, S Synthesis of benzoyl phenyl benzoates as effective inhibitors for phospholipase A2 and hyaluronidase enzymes. Bioorg Med Chem Lett15:4100-4 (2005) [PubMed] Article
Khanum, SA; Murari, SK; Vishwanth, BS; Shashikanth, S Synthesis of benzoyl phenyl benzoates as effective inhibitors for phospholipase A2 and hyaluronidase enzymes. Bioorg Med Chem Lett15:4100-4 (2005) [PubMed] Article