| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Tissue-type plasminogen activator |
|---|
| Ligand | BDBM50172832 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_320806 (CHEMBL884761) |
|---|
| Ki | 96±n/a nM |
|---|
| Citation |  Deng, JZ; McMasters, DR; Rabbat, PM; Williams, PD; Coburn, CA; Yan, Y; Kuo, LC; Lewis, SD; Lucas, BJ; Krueger, JA; Strulovici, B; Vacca, JP; Lyle, TA; Burgey, CS Development of an oxazolopyridine series of dual thrombin/factor Xa inhibitors via structure-guided lead optimization. Bioorg Med Chem Lett15:4411-6 (2005) [PubMed] Article Deng, JZ; McMasters, DR; Rabbat, PM; Williams, PD; Coburn, CA; Yan, Y; Kuo, LC; Lewis, SD; Lucas, BJ; Krueger, JA; Strulovici, B; Vacca, JP; Lyle, TA; Burgey, CS Development of an oxazolopyridine series of dual thrombin/factor Xa inhibitors via structure-guided lead optimization. Bioorg Med Chem Lett15:4411-6 (2005) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Tissue-type plasminogen activator |
|---|
| Name: | Tissue-type plasminogen activator |
|---|
| Synonyms: | Alteplase | PLAT | Reteplase | TPA_HUMAN | Thrombin receptor protein | Tissue-type plasminogen activator | Tissue-type plasminogen activator (tPA) | Tissue-type plasminogen activator precursor | t-PA | t-Plasminogen Activator (tPA) | t-plasminogen activator |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 62931.08 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | n/a |
|---|
| Residue: | 562 |
|---|
| Sequence: | MDAMKRGLCCVLLLCGAVFVSPSQEIHARFRRGARSYQVICRDEKTQMIYQQHQSWLRPV
LRSNRVEYCWCNSGRAQCHSVPVKSCSEPRCFNGGTCQQALYFSDFVCQCPEGFAGKCCE
IDTRATCYEDQGISYRGTWSTAESGAECTNWNSSALAQKPYSGRRPDAIRLGLGNHNYCR
NPDRDSKPWCYVFKAGKYSSEFCSTPACSEGNSDCYFGNGSAYRGTHSLTESGASCLPWN
SMILIGKVYTAQNPSAQALGLGKHNYCRNPDGDAKPWCHVLKNRRLTWEYCDVPSCSTCG
LRQYSQPQFRIKGGLFADIASHPWQAAIFAKHRRSPGERFLCGGILISSCWILSAAHCFQ
ERFPPHHLTVILGRTYRVVPGEEEQKFEVEKYIVHKEFDDDTYDNDIALLQLKSDSSRCA
QESSVVRTVCLPPADLQLPDWTECELSGYGKHEALSPFYSERLKEAHVRLYPSSRCTSQH
LLNRTVTDNMLCAGDTRSGGPQANLHDACQGDSGGPLVCLNDGRMTLVGIISWGLGCGQK
DVPGVYTKVTNYLDWIRDNMRP
|
|
|
|---|
| BDBM50172832 |
|---|
| n/a |
|---|
| Name | BDBM50172832 |
|---|
| Synonyms: | 2-(2-{2-[2-(5-Chloro-2-[1,2,4]triazol-1-yl-benzyl)-oxazolo[4,5-c]pyridin-4-ylamino]-ethyl}-piperidin-1-yl)-ethanol | CHEMBL197843 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C24H28ClN7O2 |
|---|
| Mol. Mass. | 481.978 |
|---|
| SMILES | OCCN1CCCCC1CCNc1nccc2oc(Cc3cc(Cl)ccc3-n3cncn3)nc12 |
|---|
| Structure | 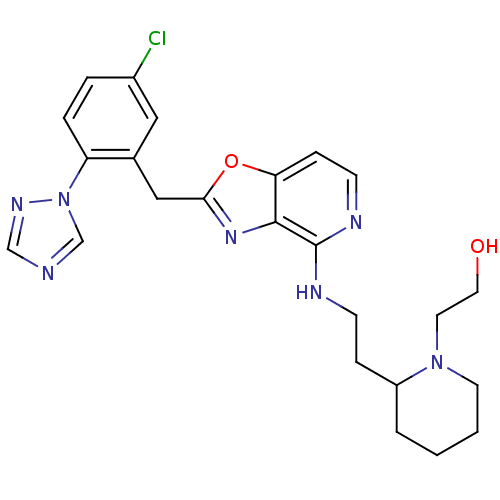
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Deng, JZ; McMasters, DR; Rabbat, PM; Williams, PD; Coburn, CA; Yan, Y; Kuo, LC; Lewis, SD; Lucas, BJ; Krueger, JA; Strulovici, B; Vacca, JP; Lyle, TA; Burgey, CS Development of an oxazolopyridine series of dual thrombin/factor Xa inhibitors via structure-guided lead optimization. Bioorg Med Chem Lett15:4411-6 (2005) [PubMed] Article
Deng, JZ; McMasters, DR; Rabbat, PM; Williams, PD; Coburn, CA; Yan, Y; Kuo, LC; Lewis, SD; Lucas, BJ; Krueger, JA; Strulovici, B; Vacca, JP; Lyle, TA; Burgey, CS Development of an oxazolopyridine series of dual thrombin/factor Xa inhibitors via structure-guided lead optimization. Bioorg Med Chem Lett15:4411-6 (2005) [PubMed] Article