| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Reverse transcriptase/RNaseH |
|---|
| Ligand | BDBM50175607 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_321370 (CHEMBL881514) |
|---|
| IC50 | 4500±n/a nM |
|---|
| Citation |  Bonache, MC; Chamorro, C; Velázquez, S; De Clercq, E; Balzarini, J; Barrios, FR; Gago, F; Camarasa, MJ; San-Félix, A Improving the antiviral efficacy and selectivity of HIV-1 reverse transcriptase inhibitor TSAO-T by the introduction of functional groups at the N-3 position. J Med Chem48:6653-60 (2005) [PubMed] Article Bonache, MC; Chamorro, C; Velázquez, S; De Clercq, E; Balzarini, J; Barrios, FR; Gago, F; Camarasa, MJ; San-Félix, A Improving the antiviral efficacy and selectivity of HIV-1 reverse transcriptase inhibitor TSAO-T by the introduction of functional groups at the N-3 position. J Med Chem48:6653-60 (2005) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Reverse transcriptase/RNaseH |
|---|
| Name: | Reverse transcriptase/RNaseH |
|---|
| Synonyms: | HIV-1 Reverse Transcriptase RNase H | Human immunodeficiency virus type 1 reverse transcriptase | Reverse transcriptase/RNaseH |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 65229.15 |
|---|
| Organism: | Human immunodeficiency virus 1 |
|---|
| Description: | ChEMBL_1473730 |
|---|
| Residue: | 566 |
|---|
| Sequence: | PISPIETVPVKLKPGMDGPKVKQWPLTEEKIKALVEICTEMEKEGKISKIGPENPYNTPV
FAIKKKDSTKWRKLVDFRELNKRTQDFWEVQLGIPHPAGLKKRKSVTVLDVGDAYFSVPL
DEDFRKYTAFTIPSINNETPGIRYQYNVLPQGWKGSPAIFQSSMTKILEPFRKQNPDIVI
YQYMDDLYVGSDLEIGQHRTKIEELRQHLLRWGLTTPDKKHQKEPPFLWMGYELHPDKWT
VQPIVLPEKDSWTVNDIQKLVGKLNWASQIYPGIRVRQLCKLLRGTKALTEVIPLTEEAE
LELAENREILKEPVHGVYYDPSKDLIAEIQKQGQGQWTYQIYQEPFKNLRTGKYARMRGA
HTNDVKQLTEAVQKITTESIVIWGKTPKFKLPIQKETWETWWTEYWQATWIPEWEFVNTP
PLVKLWYQLEKEPIVGAETFYVDGAANRETKLGKAGYVTNRGRQKVVTLTDTTNQKTELQ
AIYLALQDSGLEVNIVTDSQYALGIIQAQPDQSESELVNQIIEQLIKKEKVYLAWVPAHK
GIGGNEQVDKLVSAGIRKVLFLDGID
|
|
|
|---|
| BDBM50175607 |
|---|
| n/a |
|---|
| Name | BDBM50175607 |
|---|
| Synonyms: | 1-[4-Amino-9-(tert-butyl-dimethyl-silanyloxy)-6-(tert-butyl-dimethyl-silanyloxymethyl)-2,2-dioxo-1,7-dioxa-2lambda*6*-thia-spiro[4.4]non-3-en-8-yl]-3-(4-hydroxy-but-2-enyl)-5-methyl-1H-pyrimidine-2,4-dione | CHEMBL3143798 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C28H49N3O9SSi2 |
|---|
| Mol. Mass. | 659.939 |
|---|
| SMILES | Cc1cn([C@@H]2O[C@H](CO[Si](C)(C)C(C)(C)C)[C@@]3(OS(=O)(=O)C=C3N)[C@H]2O[Si](C)(C)C(C)(C)C)c(=O)n(C\C=C\CO)c1=O |r,c:21| |
|---|
| Structure | 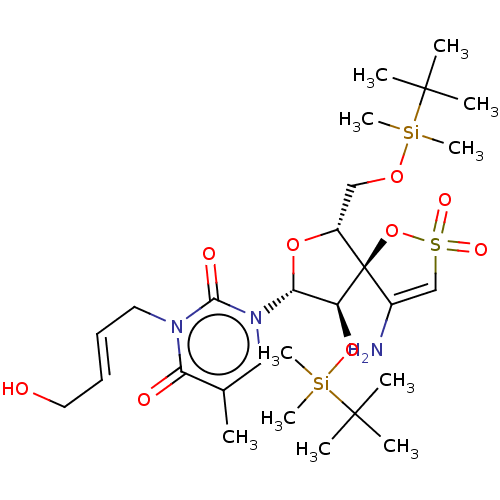
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Bonache, MC; Chamorro, C; Velázquez, S; De Clercq, E; Balzarini, J; Barrios, FR; Gago, F; Camarasa, MJ; San-Félix, A Improving the antiviral efficacy and selectivity of HIV-1 reverse transcriptase inhibitor TSAO-T by the introduction of functional groups at the N-3 position. J Med Chem48:6653-60 (2005) [PubMed] Article
Bonache, MC; Chamorro, C; Velázquez, S; De Clercq, E; Balzarini, J; Barrios, FR; Gago, F; Camarasa, MJ; San-Félix, A Improving the antiviral efficacy and selectivity of HIV-1 reverse transcriptase inhibitor TSAO-T by the introduction of functional groups at the N-3 position. J Med Chem48:6653-60 (2005) [PubMed] Article