

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Reaction Details | |||
|---|---|---|---|
 | Report a problem with these data | ||
| Target | 17-beta-hydroxysteroid dehydrogenase type 1 | ||
| Ligand | BDBM50179201 | ||
| Substrate/Competitor | n/a | ||
| Meas. Tech. | ChEMBL_325410 (CHEMBL858655) | ||
| IC50 | 52±n/a nM | ||
| Citation |  Poirier, D; Boivin, RP; Tremblay, MR; Bérubé, M; Qiu, W; Lin, SX Estradiol-adenosine hybrid compounds designed to inhibit type 1 17beta-hydroxysteroid dehydrogenase. J Med Chem48:8134-47 (2005) [PubMed] Article Poirier, D; Boivin, RP; Tremblay, MR; Bérubé, M; Qiu, W; Lin, SX Estradiol-adenosine hybrid compounds designed to inhibit type 1 17beta-hydroxysteroid dehydrogenase. J Med Chem48:8134-47 (2005) [PubMed] Article | ||
| More Info.: | Get all data from this article, Assay Method | ||
| 17-beta-hydroxysteroid dehydrogenase type 1 | |||
| Name: | 17-beta-hydroxysteroid dehydrogenase type 1 | ||
| Synonyms: | 17-beta-HSD 1 | 17-beta-Hydroxysteroid Dehydrogenase 1 (17-beta-HSD1) | 17-beta-hydroxysteroid dehydrogenase type 1 | 20 alpha-hydroxysteroid dehydrogenase | 20-alpha-HSD | DHB1_HUMAN | E17KSR | E2DH | EDH17B1 | EDH17B2 | EDHB17 | Estradiol 17-beta-dehydrogenase 1 | Estradiol 17-beta-dehydrogenase 1 (17beta-HSD1) | HSD17B1 | Placental 17-beta-hydroxysteroid dehydrogenase | SDR28C1 | ||
| Type: | Enzyme | ||
| Mol. Mass.: | 34945.13 | ||
| Organism: | Homo sapiens (Human) | ||
| Description: | P14061 | ||
| Residue: | 328 | ||
| Sequence: |
| ||
| BDBM50179201 | |||
| n/a | |||
| Name | BDBM50179201 | ||
| Synonyms: | ((2R,3S,4R,5R)-5-(6-amino-9H-purin-9-yl)-3,4-dihydroxy-tetrahydrofuran-2-yl)methyl 9-((8R,9S,13S,14S,16S,17S)-3,17-dihydroxy-13-methyl-7,8,9,11,12,13,14,15,16,17-decahydro-6H-cyclopenta[a]phenanthren-16-yl)nonanoate | ((2R,3S,4R,5R)-5-(6-amino-9H-purin-9-yl)-3,4-dihydroxytetrahydrofuran-2-yl)methyl 9-((8R,9S,13S,14S,16S,17S)-3,17-dihydroxy-13-methyl-7,8,9,11,12,13,14,15,16,17-decahydro-6H-cyclopenta[a]phenanthren-16-yl)nonanoate | 5'-O-{9-[3',17'beta-dihydroxy-1',3',5'(10')-estratrien-16'beta-yl]-nonanoyl} adenosine | CHEMBL371948 | O5'-[9-(3,17B-DIHYDROXY-1,3,5(10)-ESTRATRIEN-16B-YL)-NONANOYL]ADENOSINE | ||
| Type | Small organic molecule | ||
| Emp. Form. | C37H51N5O7 | ||
| Mol. Mass. | 677.8301 | ||
| SMILES | C[C@]12CC[C@H]3[C@@H](CCc4cc(O)ccc34)[C@@H]1C[C@H](CCCCCCCCC(=O)OC[C@H]1O[C@H]([C@H](O)[C@@H]1O)n1cnc3c(N)ncnc13)[C@@H]2O |r| | ||
| Structure | 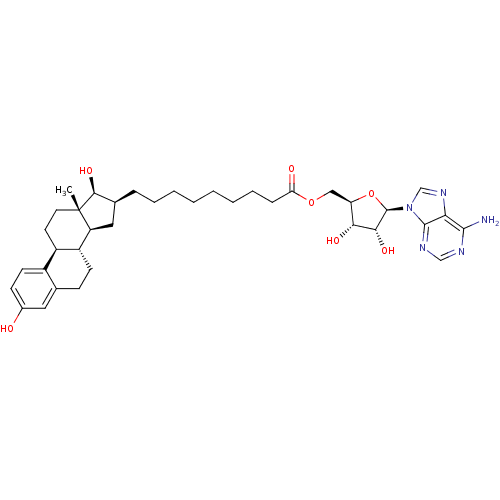
| ||