

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Reaction Details | |||
|---|---|---|---|
 | Report a problem with these data | ||
| Target | Reverse transcriptase/RNaseH | ||
| Ligand | BDBM50183203 | ||
| Substrate/Competitor | n/a | ||
| Meas. Tech. | ChEMBL_355438 (CHEMBL868998) | ||
| IC50 | 1252±n/a nM | ||
| Citation |  Muraglia, E; Kinzel, OD; Laufer, R; Miller, MD; Moyer, G; Munshi, V; Orvieto, F; Palumbi, MC; Pescatore, G; Rowley, M; Williams, PD; Summa, V Tetrazole thioacetanilides: potent non-nucleoside inhibitors of WT HIV reverse transcriptase and its K103N mutant. Bioorg Med Chem Lett16:2748-52 (2006) [PubMed] Article Muraglia, E; Kinzel, OD; Laufer, R; Miller, MD; Moyer, G; Munshi, V; Orvieto, F; Palumbi, MC; Pescatore, G; Rowley, M; Williams, PD; Summa, V Tetrazole thioacetanilides: potent non-nucleoside inhibitors of WT HIV reverse transcriptase and its K103N mutant. Bioorg Med Chem Lett16:2748-52 (2006) [PubMed] Article | ||
| More Info.: | Get all data from this article, Assay Method | ||
| Reverse transcriptase/RNaseH | |||
| Name: | Reverse transcriptase/RNaseH | ||
| Synonyms: | HIV-1 Reverse Transcriptase RNase H | Human immunodeficiency virus type 1 reverse transcriptase | Reverse transcriptase/RNaseH | ||
| Type: | PROTEIN | ||
| Mol. Mass.: | 65229.15 | ||
| Organism: | Human immunodeficiency virus 1 | ||
| Description: | ChEMBL_1473730 | ||
| Residue: | 566 | ||
| Sequence: |
| ||
| BDBM50183203 | |||
| n/a | |||
| Name | BDBM50183203 | ||
| Synonyms: | CHEMBL208008 | N-(4-sulfamoyl-phenyl)-2-[1-(2,4,6-trimethyl-phenyl)-1H-tetrazol-5-ylsulfanyl]-acetamide | ||
| Type | Small organic molecule | ||
| Emp. Form. | C18H20N6O3S2 | ||
| Mol. Mass. | 432.52 | ||
| SMILES | Cc1cc(C)c(c(C)c1)-n1nnnc1SCC(=O)Nc1ccc(cc1)S(N)(=O)=O |(13.95,-2.13,;14.28,-.63,;13.14,.41,;13.47,1.92,;12.34,2.96,;14.94,2.39,;16.08,1.35,;17.54,1.82,;15.75,-.16,;15.26,3.89,;14.24,5.05,;15.01,6.38,;16.52,6.05,;16.67,4.52,;18,3.75,;19.34,4.51,;20.67,3.74,;20.67,2.2,;22.01,4.51,;23.34,3.74,;23.33,2.2,;24.66,1.42,;26,2.19,;26,3.74,;24.66,4.5,;27.33,1.42,;28.66,.65,;26.56,.09,;28.11,2.75,)| | ||
| Structure | 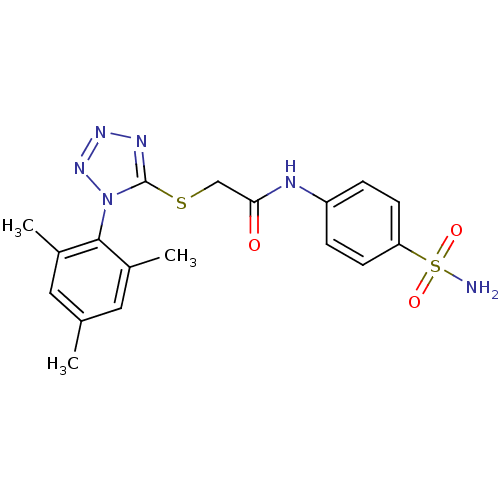
| ||