| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Calpain-1 catalytic subunit |
|---|
| Ligand | BDBM50193493 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_386232 (CHEMBL869269) |
|---|
| Ki | 196±n/a nM |
|---|
| Citation |  Korukonda, R; Guan, N; Dalton, JT; Liu, J; Donkor, IO Synthesis, calpain inhibitory activity, and cytotoxicity of P2-substituted proline and thiaproline peptidyl aldehydes and peptidyl alpha-ketoamides. J Med Chem49:5282-90 (2006) [PubMed] Article Korukonda, R; Guan, N; Dalton, JT; Liu, J; Donkor, IO Synthesis, calpain inhibitory activity, and cytotoxicity of P2-substituted proline and thiaproline peptidyl aldehydes and peptidyl alpha-ketoamides. J Med Chem49:5282-90 (2006) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Calpain-1 catalytic subunit |
|---|
| Name: | Calpain-1 catalytic subunit |
|---|
| Synonyms: | CAN1_PIG | CANP 1 | CAPN1 | Calcium-activated neutral proteinase 1 | Calpain 1 | Calpain mu-type | Calpain-1 | Calpain-1 catalytic subunit | Calpain-1 large subunit | Micromolar-calpain | muCANP |
|---|
| Type: | Catalytic subunit; forms a heterodimer with a small (regulatory) subunit (CAPNS1) |
|---|
| Mol. Mass.: | 81729.82 |
|---|
| Organism: | Sus scrofa (pig) |
|---|
| Description: | Native calpain-1 from porcine erythrocytes, purchased from Calbiochem, was used in assay. |
|---|
| Residue: | 714 |
|---|
| Sequence: | MAEEVITPVYCTGVSAQVQKLRAKELGLGRHENAIKYLGQDYEQLRAHCLQSGSLFRDEA
FPPVPQSLGFKELGPNSSKTYGVKWKRPTELFSNPQFIVDGATRTDICQGALGDCWLLAA
IASLTLNDTLLHRVVPHGQSFQNGYAGIFHFQLWQFGEWVDVVVDDLLPTKDGKLVFVHS
AQGNEFWSALLEKAYAKVNGSYEALSGGSTSEGFEDFTGGVTEWYELRKAPSDLYSIILK
ALERGSLLGCSIDISSVLDMEAVTFKKLVKGHAYSVTGAKQVNYQGQMVNLIRMRNPWGE
VEWTGAWSDGSSEWNGVDPYQRDQLRVRMEDGEFWMSFRDFLREFTRLEICNLTPDALKS
QRVRNWNTTLYEGTWRRGSTAGGCRNYPATFWVNPQFKIRLEETDDPEDDYGGRESGCSF
VLALMQKHRRRERRFGRDMETIGFAVYEVPPELVGQPVHLKRDFFLANASRARSEQFINL
REVSTRFRLPPGEYVVVPSTFEPNKEGDFVLRFFSEKKAGTQELDDQVQAILPDEQVLSE
EEIDENFKALFRQLAGEDMEISVRELRTILNRIISKHKDLRTKGFSLESCRSMVNLMDRD
GNGKLGLVEFNILWNRIRNYLSIFRKFDLDKSGSMSAYEMRMAIESAGFKLNKKLFELII
TRYSEPDLAVDFDNFVCCLVRLETMFRFFKTLDTDLDGVVTFDLFKWLQLTMFA
|
|
|
|---|
| BDBM50193493 |
|---|
| n/a |
|---|
| Name | BDBM50193493 |
|---|
| Synonyms: | (R)-N-((R,S)-1-oxo-3-phenylpropan-2-yl)-1-(phenylsulfonyl)-pyrrolidine-2-carboxamide | CHEMBL378494 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C20H22N2O4S |
|---|
| Mol. Mass. | 386.465 |
|---|
| SMILES | O=CC(Cc1ccccc1)NC(=O)[C@H]1CCCN1S(=O)(=O)c1ccccc1 |
|---|
| Structure | 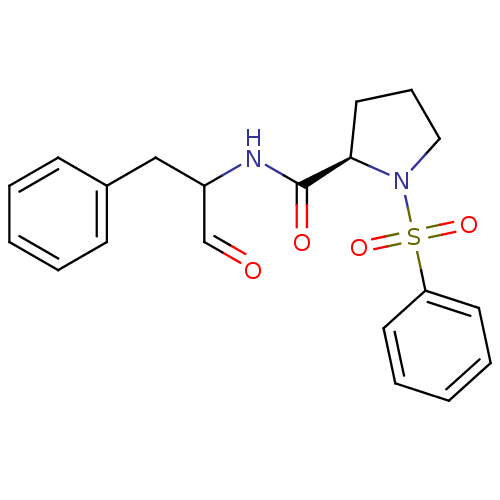
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Korukonda, R; Guan, N; Dalton, JT; Liu, J; Donkor, IO Synthesis, calpain inhibitory activity, and cytotoxicity of P2-substituted proline and thiaproline peptidyl aldehydes and peptidyl alpha-ketoamides. J Med Chem49:5282-90 (2006) [PubMed] Article
Korukonda, R; Guan, N; Dalton, JT; Liu, J; Donkor, IO Synthesis, calpain inhibitory activity, and cytotoxicity of P2-substituted proline and thiaproline peptidyl aldehydes and peptidyl alpha-ketoamides. J Med Chem49:5282-90 (2006) [PubMed] Article