| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Induced myeloid leukemia cell differentiation protein Mcl-1 |
|---|
| Ligand | BDBM50196032 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_416891 (CHEMBL909447) |
|---|
| Ki | 260±n/a nM |
|---|
| Citation |  Wang, G; Nikolovska-Coleska, Z; Yang, CY; Wang, R; Tang, G; Guo, J; Shangary, S; Qiu, S; Gao, W; Yang, D; Meagher, J; Stuckey, J; Krajewski, K; Jiang, S; Roller, PP; Abaan, HO; Tomita, Y; Wang, S Structure-based design of potent small-molecule inhibitors of anti-apoptotic Bcl-2 proteins. J Med Chem49:6139-42 (2006) [PubMed] Article Wang, G; Nikolovska-Coleska, Z; Yang, CY; Wang, R; Tang, G; Guo, J; Shangary, S; Qiu, S; Gao, W; Yang, D; Meagher, J; Stuckey, J; Krajewski, K; Jiang, S; Roller, PP; Abaan, HO; Tomita, Y; Wang, S Structure-based design of potent small-molecule inhibitors of anti-apoptotic Bcl-2 proteins. J Med Chem49:6139-42 (2006) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Induced myeloid leukemia cell differentiation protein Mcl-1 |
|---|
| Name: | Induced myeloid leukemia cell differentiation protein Mcl-1 |
|---|
| Synonyms: | BCL2L3 | Bcl-2-like protein 3 | Bcl-2-like protein 3 (Mcl-1) | Bcl-2-related protein EAT/mcl1 | Bcl2-L-3 | Induced myeloid leukemia cell differentiation protein (Mcl-1) | MCL1 | MCL1_HUMAN | Mcl-1 | Myeloid Cell factor-1 (Mcl-1) | Myeloid cell leukemia sequence 1 (BCL2-related) | mcl1/EAT |
|---|
| Type: | Membrane; Single-pass membrane protein |
|---|
| Mol. Mass.: | 37332.87 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Q07820 |
|---|
| Residue: | 350 |
|---|
| Sequence: | MFGLKRNAVIGLNLYCGGAGLGAGSGGATRPGGRLLATEKEASARREIGGGEAGAVIGGS
AGASPPSTLTPDSRRVARPPPIGAEVPDVTATPARLLFFAPTRRAAPLEEMEAPAADAIM
SPEEELDGYEPEPLGKRPAVLPLLELVGESGNNTSTDGSLPSTPPPAEEEEDELYRQSLE
IISRYLREQATGAKDTKPMGRSGATSRKALETLRRVGDGVQRNHETAFQGMLRKLDIKNE
DDVKSLSRVMIHVFSDGVTNWGRIVTLISFGAFVAKHLKTINQESCIEPLAESITDVLVR
TKRDWLVKQRGWDGFVEFFHVEDLEGGIRNVLLAFAGVAGVGAGLAYLIR
|
|
|
|---|
| BDBM50196032 |
|---|
| n/a |
|---|
| Name | BDBM50196032 |
|---|
| Synonyms: | (N-[(2-tert-butylbenzenesulfonyl)phenyl]-2,3,4-trihydroxy-5-(2-isopropylbenzyl)benzamide | CHEMBL217354 | N-[(2-tert-butyl-benzenesulfonyl)-phenyl]-2,3,4-trihydroxy-5-(2-isopropyl-benzyl)-benzamide | TW-37 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C33H35NO6S |
|---|
| Mol. Mass. | 573.699 |
|---|
| SMILES | CC(C)c1ccccc1Cc1cc(C(=O)Nc2ccc(cc2)S(=O)(=O)c2ccccc2C(C)(C)C)c(O)c(O)c1O |
|---|
| Structure | 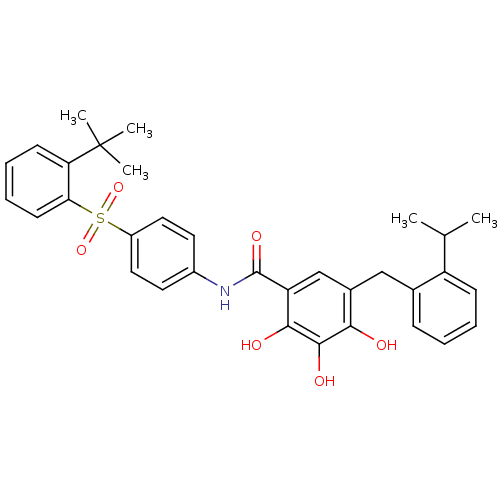
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Wang, G; Nikolovska-Coleska, Z; Yang, CY; Wang, R; Tang, G; Guo, J; Shangary, S; Qiu, S; Gao, W; Yang, D; Meagher, J; Stuckey, J; Krajewski, K; Jiang, S; Roller, PP; Abaan, HO; Tomita, Y; Wang, S Structure-based design of potent small-molecule inhibitors of anti-apoptotic Bcl-2 proteins. J Med Chem49:6139-42 (2006) [PubMed] Article
Wang, G; Nikolovska-Coleska, Z; Yang, CY; Wang, R; Tang, G; Guo, J; Shangary, S; Qiu, S; Gao, W; Yang, D; Meagher, J; Stuckey, J; Krajewski, K; Jiang, S; Roller, PP; Abaan, HO; Tomita, Y; Wang, S Structure-based design of potent small-molecule inhibitors of anti-apoptotic Bcl-2 proteins. J Med Chem49:6139-42 (2006) [PubMed] Article