| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Protein-glutamine gamma-glutamyltransferase 2 |
|---|
| Ligand | BDBM50200152 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_422560 (CHEMBL912249) |
|---|
| Ki | 1300±n/a nM |
|---|
| Citation |  Watts, RE; Siegel, M; Khosla, C Structure-activity relationship analysis of the selective inhibition of transglutaminase 2 by dihydroisoxazoles. J Med Chem49:7493-501 (2006) [PubMed] Article Watts, RE; Siegel, M; Khosla, C Structure-activity relationship analysis of the selective inhibition of transglutaminase 2 by dihydroisoxazoles. J Med Chem49:7493-501 (2006) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Protein-glutamine gamma-glutamyltransferase 2 |
|---|
| Name: | Protein-glutamine gamma-glutamyltransferase 2 |
|---|
| Synonyms: | Protein-glutamine gamma-glutamyltransferase | Protein-glutamine gamma-glutamyltransferase 2 (TG2) | TGM2 | TGM2_HUMAN | Tissue transglutaminase | Tissue transglutaminase (TG2) | Transglutaminase 2 (TGM2) | Transglutaminase C | Transglutaminase-2 |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 77309.30 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P21980 |
|---|
| Residue: | 687 |
|---|
| Sequence: | MAEELVLERCDLELETNGRDHHTADLCREKLVVRRGQPFWLTLHFEGRNYEASVDSLTFS
VVTGPAPSQEAGTKARFPLRDAVEEGDWTATVVDQQDCTLSLQLTTPANAPIGLYRLSLE
ASTGYQGSSFVLGHFILLFNAWCPADAVYLDSEEERQEYVLTQQGFIYQGSAKFIKNIPW
NFGQFEDGILDICLILLDVNPKFLKNAGRDCSRRSSPVYVGRVVSGMVNCNDDQGVLLGR
WDNNYGDGVSPMSWIGSVDILRRWKNHGCQRVKYGQCWVFAAVACTVLRCLGIPTRVVTN
YNSAHDQNSNLLIEYFRNEFGEIQGDKSEMIWNFHCWVESWMTRPDLQPGYEGWQALDPT
PQEKSEGTYCCGPVPVRAIKEGDLSTKYDAPFVFAEVNADVVDWIQQDDGSVHKSINRSL
IVGLKISTKSVGRDEREDITHTYKYPEGSSEEREAFTRANHLNKLAEKEETGMAMRIRVG
QSMNMGSDFDVFAHITNNTAEEYVCRLLLCARTVSYNGILGPECGTKYLLNLNLEPFSEK
SVPLCILYEKYRDCLTESNLIKVRALLVEPVINSYLLAERDLYLENPEIKIRILGEPKQK
RKLVAEVSLQNPLPVALEGCTFTVEGAGLTEEQKTVEIPDPVEAGEEVKVRMDLLPLHMG
LHKLVVNFESDKLKAVKGFRNVIIGPA
|
|
|
|---|
| BDBM50200152 |
|---|
| n/a |
|---|
| Name | BDBM50200152 |
|---|
| Synonyms: | CHEMBL219427 | quinolin-3-ylmethyl (S)-1-(((S)-3-bromo-4,5-dihydroisoxazol-5-yl)methylamino)-3-(5-fluoro-1H-indol-3-yl)-1-oxopropan-2-ylcarbamate |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C26H23BrFN5O4 |
|---|
| Mol. Mass. | 568.394 |
|---|
| SMILES | Fc1ccc2[nH]cc(C[C@H](NC(=O)OCc3cnc4ccccc4c3)C(=O)NC[C@@H]3CC(Br)=NO3)c2c1 |r,c:34| |
|---|
| Structure | 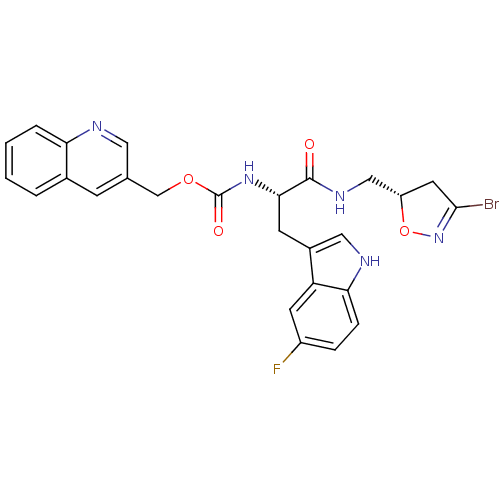
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Watts, RE; Siegel, M; Khosla, C Structure-activity relationship analysis of the selective inhibition of transglutaminase 2 by dihydroisoxazoles. J Med Chem49:7493-501 (2006) [PubMed] Article
Watts, RE; Siegel, M; Khosla, C Structure-activity relationship analysis of the selective inhibition of transglutaminase 2 by dihydroisoxazoles. J Med Chem49:7493-501 (2006) [PubMed] Article