| Reaction Details |
|---|
 | Report a problem with these data |
| Target | B2 bradykinin receptor |
|---|
| Ligand | BDBM50203200 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_425212 (CHEMBL855158) |
|---|
| Ki | >50000±n/a nM |
|---|
| Citation |  D'Amico, DC; Aya, T; Human, J; Fotsch, C; Chen, JJ; Biswas, K; Riahi, B; Norman, MH; Willoughby, CA; Hungate, R; Reider, PJ; Biddlecome, G; Lester-Zeiner, D; Staden, CV; Johnson, E; Kamassah, A; Arik, L; Wang, J; Viswanadhan, VN; Groneberg, RD; Zhan, J; Suzuki, H; Toro, A; Mareska, DA; Clarke, DE; Harvey, DM; Burgess, LE; Laird, ER; Askew, B; Ng, G Identification of a nonpeptidic and conformationally restricted bradykinin B1 receptor antagonist with anti-inflammatory activity. J Med Chem50:607-10 (2007) [PubMed] Article D'Amico, DC; Aya, T; Human, J; Fotsch, C; Chen, JJ; Biswas, K; Riahi, B; Norman, MH; Willoughby, CA; Hungate, R; Reider, PJ; Biddlecome, G; Lester-Zeiner, D; Staden, CV; Johnson, E; Kamassah, A; Arik, L; Wang, J; Viswanadhan, VN; Groneberg, RD; Zhan, J; Suzuki, H; Toro, A; Mareska, DA; Clarke, DE; Harvey, DM; Burgess, LE; Laird, ER; Askew, B; Ng, G Identification of a nonpeptidic and conformationally restricted bradykinin B1 receptor antagonist with anti-inflammatory activity. J Med Chem50:607-10 (2007) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| B2 bradykinin receptor |
|---|
| Name: | B2 bradykinin receptor |
|---|
| Synonyms: | B2 BRADYKININ | B2 bradykinin receptor | B2R | BDKRB2 | BK-2 receptor | BKR2 | BKRB2_HUMAN | Bradykinin B2 receptor |
|---|
| Type: | Enzyme Catalytic Domain |
|---|
| Mol. Mass.: | 44467.17 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | B2 BRADYKININ BDKRB2 HUMAN::P30411 |
|---|
| Residue: | 391 |
|---|
| Sequence: | MFSPWKISMFLSVREDSVPTTASFSADMLNVTLQGPTLNGTFAQSKCPQVEWLGWLNTIQ
PPFLWVLFVLATLENIFVLSVFCLHKSSCTVAEIYLGNLAAADLILACGLPFWAITISNN
FDWLFGETLCRVVNAIISMNLYSSICFLMLVSIDRYLALVKTMSMGRMRGVRWAKLYSLV
IWGCTLLLSSPMLVFRTMKEYSDEGHNVTACVISYPSLIWEVFTNMLLNVVGFLLPLSVI
TFCTMQIMQVLRNNEMQKFKEIQTERRATVLVLVVLLLFIICWLPFQISTFLDTLHRLGI
LSSCQDERIIDVITQIASFMAYSNSCLNPLVYVIVGKRFRKKSWEVYQGVCQKGGCRSEP
IQMENSMGTLRTSISVERQIHKLQDWAGSRQ
|
|
|
|---|
| BDBM50203200 |
|---|
| n/a |
|---|
| Name | BDBM50203200 |
|---|
| Synonyms: | (R)-3-(naphthalene-7-sulfonamido)-3-phenyl-N-((R)-7-(piperidin-1-ylmethyl)-3,4-dihydro-2H-chromen-4-yl)propanamide | (R)-3-(naphthalene-7-sulfonamido)-3-phenyl-N-((R)-7-(piperidin-1-ylmethyl)chroman-4-yl)propanamide | CHEMBL415848 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C34H37N3O4S |
|---|
| Mol. Mass. | 583.74 |
|---|
| SMILES | O=C(C[C@@H](NS(=O)(=O)c1ccc2ccccc2c1)c1ccccc1)N[C@@H]1CCOc2cc(CN3CCCCC3)ccc12 |
|---|
| Structure | 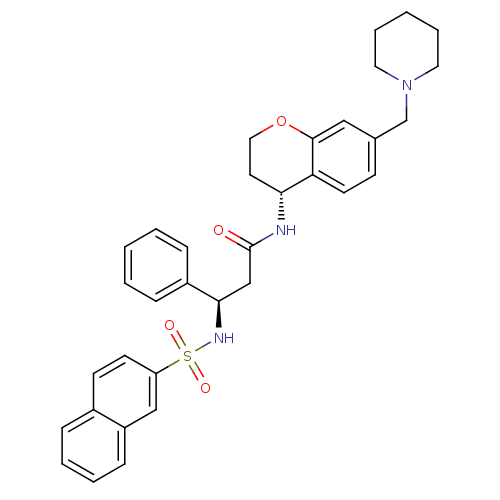
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 D'Amico, DC; Aya, T; Human, J; Fotsch, C; Chen, JJ; Biswas, K; Riahi, B; Norman, MH; Willoughby, CA; Hungate, R; Reider, PJ; Biddlecome, G; Lester-Zeiner, D; Staden, CV; Johnson, E; Kamassah, A; Arik, L; Wang, J; Viswanadhan, VN; Groneberg, RD; Zhan, J; Suzuki, H; Toro, A; Mareska, DA; Clarke, DE; Harvey, DM; Burgess, LE; Laird, ER; Askew, B; Ng, G Identification of a nonpeptidic and conformationally restricted bradykinin B1 receptor antagonist with anti-inflammatory activity. J Med Chem50:607-10 (2007) [PubMed] Article
D'Amico, DC; Aya, T; Human, J; Fotsch, C; Chen, JJ; Biswas, K; Riahi, B; Norman, MH; Willoughby, CA; Hungate, R; Reider, PJ; Biddlecome, G; Lester-Zeiner, D; Staden, CV; Johnson, E; Kamassah, A; Arik, L; Wang, J; Viswanadhan, VN; Groneberg, RD; Zhan, J; Suzuki, H; Toro, A; Mareska, DA; Clarke, DE; Harvey, DM; Burgess, LE; Laird, ER; Askew, B; Ng, G Identification of a nonpeptidic and conformationally restricted bradykinin B1 receptor antagonist with anti-inflammatory activity. J Med Chem50:607-10 (2007) [PubMed] Article