| Reaction Details |
|---|
 | Report a problem with these data |
| Target | 5-hydroxytryptamine receptor 2A |
|---|
| Ligand | BDBM50209974 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_458978 (CHEMBL925072) |
|---|
| IC50 | 5300±n/a nM |
|---|
| Citation |  Pruitt, JR; Batt, DG; Wacker, DA; Bostrom, LL; Booker, SK; McLaughlin, E; Houghton, GC; Varnes, JG; Christ, DD; Covington, M; Das, AM; Davies, P; Graden, D; Kariv, I; Orlovsky, Y; Stowell, NC; Vaddi, KG; Wadman, EA; Welch, PK; Yeleswaram, S; Solomon, KA; Newton, RC; Decicco, CP; Carter, PH; Ko, SS CC chemokine receptor-3 (CCR3) antagonists: improving the selectivity of DPC168 by reducing central ring lipophilicity. Bioorg Med Chem Lett17:2992-7 (2007) [PubMed] Article Pruitt, JR; Batt, DG; Wacker, DA; Bostrom, LL; Booker, SK; McLaughlin, E; Houghton, GC; Varnes, JG; Christ, DD; Covington, M; Das, AM; Davies, P; Graden, D; Kariv, I; Orlovsky, Y; Stowell, NC; Vaddi, KG; Wadman, EA; Welch, PK; Yeleswaram, S; Solomon, KA; Newton, RC; Decicco, CP; Carter, PH; Ko, SS CC chemokine receptor-3 (CCR3) antagonists: improving the selectivity of DPC168 by reducing central ring lipophilicity. Bioorg Med Chem Lett17:2992-7 (2007) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| 5-hydroxytryptamine receptor 2A |
|---|
| Name: | 5-hydroxytryptamine receptor 2A |
|---|
| Synonyms: | 5-HT-2 | 5-HT-2A | 5-HT2A | 5-hydroxytryptamine receptor 2A (5-HT-2A) | 5-hydroxytryptamine receptor 2A (5HT-2A) | 5-hydroxytryptamine receptor 2A (5HT2A) | 5HT2A_HUMAN | HTR2 | HTR2A | Serotonin receptor 2A |
|---|
| Type: | undefined |
|---|
| Mol. Mass.: | 52607.65 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P28223 |
|---|
| Residue: | 471 |
|---|
| Sequence: | MDILCEENTSLSSTTNSLMQLNDDTRLYSNDFNSGEANTSDAFNWTVDSENRTNLSCEGC
LSPSCLSLLHLQEKNWSALLTAVVIILTIAGNILVIMAVSLEKKLQNATNYFLMSLAIAD
MLLGFLVMPVSMLTILYGYRWPLPSKLCAVWIYLDVLFSTASIMHLCAISLDRYVAIQNP
IHHSRFNSRTKAFLKIIAVWTISVGISMPIPVFGLQDDSKVFKEGSCLLADDNFVLIGSF
VSFFIPLTIMVITYFLTIKSLQKEATLCVSDLGTRAKLASFSFLPQSSLSSEKLFQRSIH
REPGSYTGRRTMQSISNEQKACKVLGIVFFLFVVMWCPFFITNIMAVICKESCNEDVIGA
LLNVFVWIGYLSSAVNPLVYTLFNKTYRSAFSRYIQCQYKENKKPLQLILVNTIPALAYK
SSQLQMGQKKNSKQDAKTTDNDCSMVALGKQHSEEASKDNSDGVNEKVSCV
|
|
|
|---|
| BDBM50209974 |
|---|
| n/a |
|---|
| Name | BDBM50209974 |
|---|
| Synonyms: | 1-((3S,4S)-4-(((S)-3-(4-fluorobenzyl)piperidin-1-yl)methyl)-1-acetylpiperidin-3-yl)-3-(5-acetyl-4-methylthiazol-2-yl)urea | CHEMBL249854 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C27H36FN5O3S |
|---|
| Mol. Mass. | 529.67 |
|---|
| SMILES | CC(=O)N1CC[C@@H](CN2CCC[C@@H](Cc3ccc(F)cc3)C2)[C@@H](C1)NC(=O)Nc1nc(C)c(s1)C(C)=O |
|---|
| Structure | 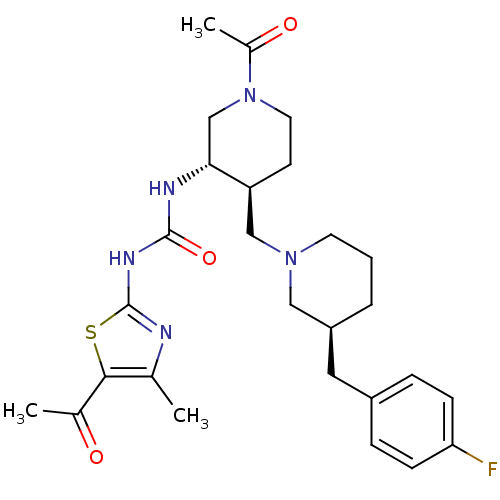
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Pruitt, JR; Batt, DG; Wacker, DA; Bostrom, LL; Booker, SK; McLaughlin, E; Houghton, GC; Varnes, JG; Christ, DD; Covington, M; Das, AM; Davies, P; Graden, D; Kariv, I; Orlovsky, Y; Stowell, NC; Vaddi, KG; Wadman, EA; Welch, PK; Yeleswaram, S; Solomon, KA; Newton, RC; Decicco, CP; Carter, PH; Ko, SS CC chemokine receptor-3 (CCR3) antagonists: improving the selectivity of DPC168 by reducing central ring lipophilicity. Bioorg Med Chem Lett17:2992-7 (2007) [PubMed] Article
Pruitt, JR; Batt, DG; Wacker, DA; Bostrom, LL; Booker, SK; McLaughlin, E; Houghton, GC; Varnes, JG; Christ, DD; Covington, M; Das, AM; Davies, P; Graden, D; Kariv, I; Orlovsky, Y; Stowell, NC; Vaddi, KG; Wadman, EA; Welch, PK; Yeleswaram, S; Solomon, KA; Newton, RC; Decicco, CP; Carter, PH; Ko, SS CC chemokine receptor-3 (CCR3) antagonists: improving the selectivity of DPC168 by reducing central ring lipophilicity. Bioorg Med Chem Lett17:2992-7 (2007) [PubMed] Article