| Reaction Details |
|---|
 | Report a problem with these data |
| Target | 5-hydroxytryptamine receptor 2A |
|---|
| Ligand | BDBM50048803 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_461981 (CHEMBL945769) |
|---|
| Ki | 0.08±n/a nM |
|---|
| Citation |  Graham, JM; Coughenour, LL; Barr, BM; Rock, DL; Nikam, SS 1-Aminoindanes as novel motif with potential atypical antipsychotic properties. Bioorg Med Chem Lett18:489-93 (2008) [PubMed] Article Graham, JM; Coughenour, LL; Barr, BM; Rock, DL; Nikam, SS 1-Aminoindanes as novel motif with potential atypical antipsychotic properties. Bioorg Med Chem Lett18:489-93 (2008) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| 5-hydroxytryptamine receptor 2A |
|---|
| Name: | 5-hydroxytryptamine receptor 2A |
|---|
| Synonyms: | 5-HT-2 | 5-HT-2A | 5-HT2A | 5-hydroxytryptamine receptor 2A (5-HT-2A) | 5-hydroxytryptamine receptor 2A (5HT-2A) | 5-hydroxytryptamine receptor 2A (5HT2A) | 5HT2A_HUMAN | HTR2 | HTR2A | Serotonin receptor 2A |
|---|
| Type: | undefined |
|---|
| Mol. Mass.: | 52607.65 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P28223 |
|---|
| Residue: | 471 |
|---|
| Sequence: | MDILCEENTSLSSTTNSLMQLNDDTRLYSNDFNSGEANTSDAFNWTVDSENRTNLSCEGC
LSPSCLSLLHLQEKNWSALLTAVVIILTIAGNILVIMAVSLEKKLQNATNYFLMSLAIAD
MLLGFLVMPVSMLTILYGYRWPLPSKLCAVWIYLDVLFSTASIMHLCAISLDRYVAIQNP
IHHSRFNSRTKAFLKIIAVWTISVGISMPIPVFGLQDDSKVFKEGSCLLADDNFVLIGSF
VSFFIPLTIMVITYFLTIKSLQKEATLCVSDLGTRAKLASFSFLPQSSLSSEKLFQRSIH
REPGSYTGRRTMQSISNEQKACKVLGIVFFLFVVMWCPFFITNIMAVICKESCNEDVIGA
LLNVFVWIGYLSSAVNPLVYTLFNKTYRSAFSRYIQCQYKENKKPLQLILVNTIPALAYK
SSQLQMGQKKNSKQDAKTTDNDCSMVALGKQHSEEASKDNSDGVNEKVSCV
|
|
|
|---|
| BDBM50048803 |
|---|
| n/a |
|---|
| Name | BDBM50048803 |
|---|
| Synonyms: | 5-(2-(4-(benzo[d]isothiazol-3-yl)piperazin-1-yl)ethyl)-6-chloroindolin-2-one | 5-[2-(4-Benzo[d]isothiazol-3-yl-piperazin-1-yl)-ethyl]-6-chloro-1,3-dihydro-indol-2-one | 5-[2-(4-Benzo[d]isothiazol-3-yl-piperazin-1-yl)-ethyl]-6-chloro-1,3-dihydro-indol-2-one (Ziprasidone) | 5-[2-(4-Benzo[d]isothiazol-3-yl-piperazin-1-yl)-ethyl]-6-chloro-1,3-dihydro-indol-2-one(Norastemizole) | CHEMBL708 | GEODON | ZIPRASIDONE | ZIPRASIDONE HYDROCHLORIDE |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C21H21ClN4OS |
|---|
| Mol. Mass. | 412.936 |
|---|
| SMILES | Clc1cc2NC(=O)Cc2cc1CCN1CCN(CC1)c1nsc2ccccc12 |
|---|
| Structure | 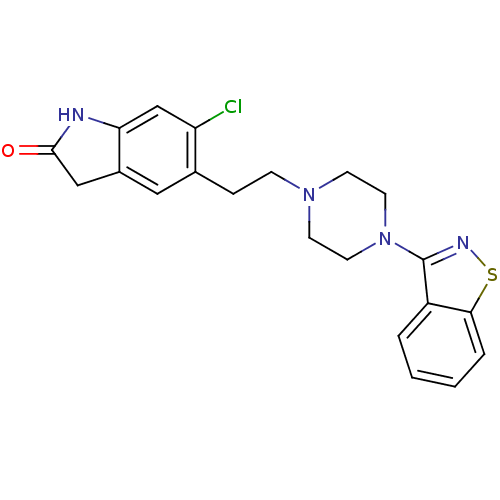
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Graham, JM; Coughenour, LL; Barr, BM; Rock, DL; Nikam, SS 1-Aminoindanes as novel motif with potential atypical antipsychotic properties. Bioorg Med Chem Lett18:489-93 (2008) [PubMed] Article
Graham, JM; Coughenour, LL; Barr, BM; Rock, DL; Nikam, SS 1-Aminoindanes as novel motif with potential atypical antipsychotic properties. Bioorg Med Chem Lett18:489-93 (2008) [PubMed] Article