| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Protein kinase C alpha type |
|---|
| Ligand | BDBM21898 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_529515 (CHEMBL966659) |
|---|
| IC50 | >100000±n/a nM |
|---|
| Citation |  Hall, JP; Kurdi, Y; Hsu, S; Cuozzo, J; Liu, J; Telliez, JB; Seidl, KJ; Winkler, A; Hu, Y; Green, N; Askew, GR; Tam, S; Clark, JD; Lin, LL Pharmacologic inhibition of tpl2 blocks inflammatory responses in primary human monocytes, synoviocytes, and blood. J Biol Chem282:33295-304 (2007) [PubMed] Article Hall, JP; Kurdi, Y; Hsu, S; Cuozzo, J; Liu, J; Telliez, JB; Seidl, KJ; Winkler, A; Hu, Y; Green, N; Askew, GR; Tam, S; Clark, JD; Lin, LL Pharmacologic inhibition of tpl2 blocks inflammatory responses in primary human monocytes, synoviocytes, and blood. J Biol Chem282:33295-304 (2007) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Protein kinase C alpha type |
|---|
| Name: | Protein kinase C alpha type |
|---|
| Synonyms: | KPCA_HUMAN | PKC alpha | PKC alpha and beta-2 | PKC-A | PKC-alpha | PKCA | PRKACA | PRKCA | Protein Kinase C- alpha | Protein kinase C alpha | Protein kinase C alpha (PKCa) | Protein kinase C alpha type (PKC-A) | Protein kinase C alpha type (PRKCA) | Proto-oncogene c-RAF | RAF proto-oncogene serine/threonine-protein kinase |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 76755.65 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | The recombinant human PKC enzymes were
produced using a baculovirus expression system in SF9 cells |
|---|
| Residue: | 672 |
|---|
| Sequence: | MADVFPGNDSTASQDVANRFARKGALRQKNVHEVKDHKFIARFFKQPTFCSHCTDFIWGF
GKQGFQCQVCCFVVHKRCHEFVTFSCPGADKGPDTDDPRSKHKFKIHTYGSPTFCDHCGS
LLYGLIHQGMKCDTCDMNVHKQCVINVPSLCGMDHTEKRGRIYLKAEVADEKLHVTVRDA
KNLIPMDPNGLSDPYVKLKLIPDPKNESKQKTKTIRSTLNPQWNESFTFKLKPSDKDRRL
SVEIWDWDRTTRNDFMGSLSFGVSELMKMPASGWYKLLNQEEGEYYNVPIPEGDEEGNME
LRQKFEKAKLGPAGNKVISPSEDRKQPSNNLDRVKLTDFNFLMVLGKGSFGKVMLADRKG
TEELYAIKILKKDVVIQDDDVECTMVEKRVLALLDKPPFLTQLHSCFQTVDRLYFVMEYV
NGGDLMYHIQQVGKFKEPQAVFYAAEISIGLFFLHKRGIIYRDLKLDNVMLDSEGHIKIA
DFGMCKEHMMDGVTTRTFCGTPDYIAPEIIAYQPYGKSVDWWAYGVLLYEMLAGQPPFDG
EDEDELFQSIMEHNVSYPKSLSKEAVSVCKGLMTKHPAKRLGCGPEGERDVREHAFFRRI
DWEKLENREIQPPFKPKVCGKGAENFDKFFTRGQPVLTPPDQLVIANIDQSDFEGFSYVN
PQFVHPILQSAV
|
|
|
|---|
| BDBM21898 |
|---|
| n/a |
|---|
| Name | BDBM21898 |
|---|
| Synonyms: | 8-chloro-4-[(3-chloro-4-fluorophenyl)amino]-6-[(1H-imidazol-4-ylmethyl)amino]quinoline-3-carbonitrile | CHEMBL436817 | Cyanoquinoline, 11 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C20H13Cl2FN6 |
|---|
| Mol. Mass. | 427.262 |
|---|
| SMILES | Fc1ccc(Nc2c(cnc3c(Cl)cc(NCc4cnc[nH]4)cc23)C#N)cc1Cl |
|---|
| Structure | 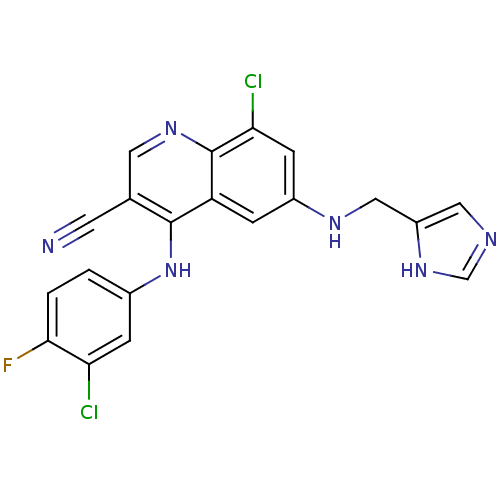
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Hall, JP; Kurdi, Y; Hsu, S; Cuozzo, J; Liu, J; Telliez, JB; Seidl, KJ; Winkler, A; Hu, Y; Green, N; Askew, GR; Tam, S; Clark, JD; Lin, LL Pharmacologic inhibition of tpl2 blocks inflammatory responses in primary human monocytes, synoviocytes, and blood. J Biol Chem282:33295-304 (2007) [PubMed] Article
Hall, JP; Kurdi, Y; Hsu, S; Cuozzo, J; Liu, J; Telliez, JB; Seidl, KJ; Winkler, A; Hu, Y; Green, N; Askew, GR; Tam, S; Clark, JD; Lin, LL Pharmacologic inhibition of tpl2 blocks inflammatory responses in primary human monocytes, synoviocytes, and blood. J Biol Chem282:33295-304 (2007) [PubMed] Article