| Reaction Details |
|---|
 | Report a problem with these data |
| Target | 5-hydroxytryptamine receptor 1A |
|---|
| Ligand | BDBM50243700 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_554184 (CHEMBL963641) |
|---|
| Ki | 2.8±n/a nM |
|---|
| Citation |  Pelletier, JC; Chengalvala, M; Cottom, J; Feingold, I; Garrick, L; Green, D; Hauze, D; Huselton, C; Jetter, J; Kao, W; Kopf, GS; Lundquist, JT; Mann, C; Mehlmann, J; Rogers, J; Shanno, L; Wrobel, J 2-phenyl-4-piperazinylbenzimidazoles: orally active inhibitors of the gonadotropin releasing hormone (GnRH) receptor. Bioorg Med Chem16:6617-40 (2008) [PubMed] Article Pelletier, JC; Chengalvala, M; Cottom, J; Feingold, I; Garrick, L; Green, D; Hauze, D; Huselton, C; Jetter, J; Kao, W; Kopf, GS; Lundquist, JT; Mann, C; Mehlmann, J; Rogers, J; Shanno, L; Wrobel, J 2-phenyl-4-piperazinylbenzimidazoles: orally active inhibitors of the gonadotropin releasing hormone (GnRH) receptor. Bioorg Med Chem16:6617-40 (2008) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| 5-hydroxytryptamine receptor 1A |
|---|
| Name: | 5-hydroxytryptamine receptor 1A |
|---|
| Synonyms: | 5-HT-1A | 5-HT1A | 5-hydroxytryptamine receptor 1A (5-HT-1A) | 5HT1A_HUMAN | ADRB2RL1 | ADRBRL1 | Dopamine D2 receptor and serotonin 1a receptor | G-21 | HTR1A | Serotonin receptor 1A |
|---|
| Type: | n/a |
|---|
| Mol. Mass.: | 46122.49 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | n/a |
|---|
| Residue: | 422 |
|---|
| Sequence: | MDVLSPGQGNNTTSPPAPFETGGNTTGISDVTVSYQVITSLLLGTLIFCAVLGNACVVAA
IALERSLQNVANYLIGSLAVTDLMVSVLVLPMAALYQVLNKWTLGQVTCDLFIALDVLCC
TSSILHLCAIALDRYWAITDPIDYVNKRTPRRAAALISLTWLIGFLISIPPMLGWRTPED
RSDPDACTISKDHGYTIYSTFGAFYIPLLLMLVLYGRIFRAARFRIRKTVKKVEKTGADT
RHGASPAPQPKKSVNGESGSRNWRLGVESKAGGALCANGAVRQGDDGAALEVIEVHRVGN
SKEHLPLPSEAGPTPCAPASFERKNERNAEAKRKMALARERKTVKTLGIIMGTFILCWLP
FFIVALVLPFCESSCHMPTLLGAIINWLGYSNSLLNPVIYAYFNKDFQNAFKKIIKCKFC
RQ
|
|
|
|---|
| BDBM50243700 |
|---|
| n/a |
|---|
| Name | BDBM50243700 |
|---|
| Synonyms: | 4-(2-(4-(2-(trifluoromethyl)-1H-benzo[d]imidazol-4-yl)piperazin-1-yl)ethoxy)-1H-benzo[d]imidazole-2(3H)-thione | CHEMBL512893 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C21H21F3N6OS |
|---|
| Mol. Mass. | 462.491 |
|---|
| SMILES | FC(F)(F)c1nc2c(cccc2[nH]1)N1CCN(CCOc2cccc3[nH]c(=S)[nH]c23)CC1 |
|---|
| Structure | 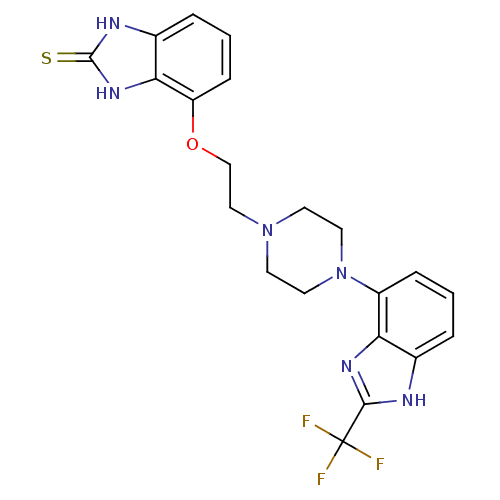
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Pelletier, JC; Chengalvala, M; Cottom, J; Feingold, I; Garrick, L; Green, D; Hauze, D; Huselton, C; Jetter, J; Kao, W; Kopf, GS; Lundquist, JT; Mann, C; Mehlmann, J; Rogers, J; Shanno, L; Wrobel, J 2-phenyl-4-piperazinylbenzimidazoles: orally active inhibitors of the gonadotropin releasing hormone (GnRH) receptor. Bioorg Med Chem16:6617-40 (2008) [PubMed] Article
Pelletier, JC; Chengalvala, M; Cottom, J; Feingold, I; Garrick, L; Green, D; Hauze, D; Huselton, C; Jetter, J; Kao, W; Kopf, GS; Lundquist, JT; Mann, C; Mehlmann, J; Rogers, J; Shanno, L; Wrobel, J 2-phenyl-4-piperazinylbenzimidazoles: orally active inhibitors of the gonadotropin releasing hormone (GnRH) receptor. Bioorg Med Chem16:6617-40 (2008) [PubMed] Article