| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Adenosine receptor A2a |
|---|
| Ligand | BDBM50243661 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_491362 (CHEMBL986416) |
|---|
| Ki | 1.1±n/a nM |
|---|
| Citation |  Shah, U; Lankin, CM; Boyle, CD; Chackalamannil, S; Greenlee, WJ; Neustadt, BR; Cohen-Williams, ME; Higgins, GA; Ng, K; Varty, GB; Zhang, H; Lachowicz, JE Design, synthesis, and evaluation of fused heterocyclic analogs of SCH 58261 as adenosine A2A receptor antagonists. Bioorg Med Chem Lett18:4204-9 (2008) [PubMed] Article Shah, U; Lankin, CM; Boyle, CD; Chackalamannil, S; Greenlee, WJ; Neustadt, BR; Cohen-Williams, ME; Higgins, GA; Ng, K; Varty, GB; Zhang, H; Lachowicz, JE Design, synthesis, and evaluation of fused heterocyclic analogs of SCH 58261 as adenosine A2A receptor antagonists. Bioorg Med Chem Lett18:4204-9 (2008) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Adenosine receptor A2a |
|---|
| Name: | Adenosine receptor A2a |
|---|
| Synonyms: | A2A adenosine receptor (hA2A) | AA2AR_HUMAN | ADENOSINE A2 | ADENOSINE A2a | ADORA2 | ADORA2A | Adenosine A2A receptor (A2AAR) |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 44716.46 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P29274 |
|---|
| Residue: | 412 |
|---|
| Sequence: | MPIMGSSVYITVELAIAVLAILGNVLVCWAVWLNSNLQNVTNYFVVSLAAADIAVGVLAI
PFAITISTGFCAACHGCLFIACFVLVLTQSSIFSLLAIAIDRYIAIRIPLRYNGLVTGTR
AKGIIAICWVLSFAIGLTPMLGWNNCGQPKEGKNHSQGCGEGQVACLFEDVVPMNYMVYF
NFFACVLVPLLLMLGVYLRIFLAARRQLKQMESQPLPGERARSTLQKEVHAAKSLAIIVG
LFALCWLPLHIINCFTFFCPDCSHAPLWLMYLAIVLSHTNSVVNPFIYAYRIREFRQTFR
KIIRSHVLRQQEPFKAAGTSARVLAAHGSDGEQVSLRLNGHPPGVWANGSAPHPERRPNG
YALGLVSGGSAQESQGNTGLPDVELLSHELKGVCPEPPGLDDPLAQDGAGVS
|
|
|
|---|
| BDBM50243661 |
|---|
| n/a |
|---|
| Name | BDBM50243661 |
|---|
| Synonyms: | 7-[2-(7-Bromo-1,2,4,5-tetrahydro-benzo[d]azepin-3-yl)-ethyl]-2-furan-2-yl-7H-pyrazolo[4,3-e][1,2,4]triazolo[1,5-c]pyrimidin-5-ylamine | CHEMBL472112 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C22H21BrN8O |
|---|
| Mol. Mass. | 493.359 |
|---|
| SMILES | Nc1nc2n(CCN3CCc4ccc(Br)cc4CC3)ncc2c2nc(nn12)-c1ccco1 |
|---|
| Structure | 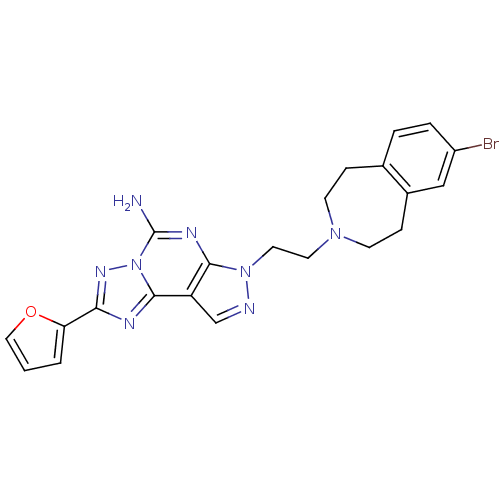
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Shah, U; Lankin, CM; Boyle, CD; Chackalamannil, S; Greenlee, WJ; Neustadt, BR; Cohen-Williams, ME; Higgins, GA; Ng, K; Varty, GB; Zhang, H; Lachowicz, JE Design, synthesis, and evaluation of fused heterocyclic analogs of SCH 58261 as adenosine A2A receptor antagonists. Bioorg Med Chem Lett18:4204-9 (2008) [PubMed] Article
Shah, U; Lankin, CM; Boyle, CD; Chackalamannil, S; Greenlee, WJ; Neustadt, BR; Cohen-Williams, ME; Higgins, GA; Ng, K; Varty, GB; Zhang, H; Lachowicz, JE Design, synthesis, and evaluation of fused heterocyclic analogs of SCH 58261 as adenosine A2A receptor antagonists. Bioorg Med Chem Lett18:4204-9 (2008) [PubMed] Article