| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Lysine--tRNA ligase |
|---|
| Ligand | BDBM50249539 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_490569 (CHEMBL981176) |
|---|
| IC50 | 4±n/a nM |
|---|
| Citation |  Hirabayashi, A; Mukaiyama, H; Kobayashi, H; Shiohara, H; Nakayama, S; Ozawa, M; Miyazawa, K; Misawa, K; Ohnota, H; Isaji, M A novel Syk family kinase inhibitor: design, synthesis, and structure-activity relationship of 1,2,4-triazolo[4,3-c]pyrimidine and 1,2,4-triazolo[1,5-c]pyrimidine derivatives. Bioorg Med Chem16:7347-57 (2008) [PubMed] Article Hirabayashi, A; Mukaiyama, H; Kobayashi, H; Shiohara, H; Nakayama, S; Ozawa, M; Miyazawa, K; Misawa, K; Ohnota, H; Isaji, M A novel Syk family kinase inhibitor: design, synthesis, and structure-activity relationship of 1,2,4-triazolo[4,3-c]pyrimidine and 1,2,4-triazolo[1,5-c]pyrimidine derivatives. Bioorg Med Chem16:7347-57 (2008) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Lysine--tRNA ligase |
|---|
| Name: | Lysine--tRNA ligase |
|---|
| Synonyms: | KARS | KARS1 | KIAA0070 | Lysyl-tRNA synthetase | SYK_HUMAN |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 68044.78 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | ChEMBL_960665 |
|---|
| Residue: | 597 |
|---|
| Sequence: | MAAVQAAEVKVDGSEPKLSKNELKRRLKAEKKVAEKEAKQKELSEKQLSQATAAATNHTT
DNGVGPEEESVDPNQYYKIRSQAIHQLKVNGEDPYPHKFHVDISLTDFIQKYSHLQPGDH
LTDITLKVAGRIHAKRASGGKLIFYDLRGEGVKLQVMANSRNYKSEEEFIHINNKLRRGD
IIGVQGNPGKTKKGELSIIPYEITLLSPCLHMLPHLHFGLKDKETRYRQRYLDLILNDFV
RQKFIIRSKIITYIRSFLDELGFLEIETPMMNIIPGGAVAKPFITYHNELDMNLYMRIAP
ELYHKMLVVGGIDRVYEIGRQFRNEGIDLTHNPEFTTCEFYMAYADYHDLMEITEKMVSG
MVKHITGSYKVTYHPDGPEGQAYDVDFTPPFRRINMVEELEKALGMKLPETNLFETEETR
KILDDICVAKAVECPPPRTTARLLDKLVGEFLEVTCINPTFICDHPQIMSPLAKWHRSKE
GLTERFELFVMKKEICNAYTELNDPMRQRQLFEEQAKAKAAGDDEAMFIDENFCTALEYG
LPPTAGWGMGIDRVAMFLTDSNNIKEVLLFPAMKPEDKKENVATTDTLESTTVGTSV
|
|
|
|---|
| BDBM50249539 |
|---|
| n/a |
|---|
| Name | BDBM50249539 |
|---|
| Synonyms: | 5-((1S,2R)-2-aminocyclohexylamino)-7-(3,5-dimethoxyphenylamino)-[1,2,4]triazolo[1,5-c]pyrimidine-8-carboxamide | 5-((1S,2R)-2-aminocyclohexylamino)-7-(3,5-dimethoxyphenylamino)-[1,2,4]triazolo[1,5-f]pyrimidine-8-carboxamide | CHEMBL475575 | cis-5-(2-Aminocyclohexylamino)-7-(3,5-dimethoxyphenylamino)-1,2,4-triazolo[1,5-c]pyrimidine-8-carboxamide |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C20H26N8O3 |
|---|
| Mol. Mass. | 426.4722 |
|---|
| SMILES | COc1cc(Nc2nc(N[C@H]3CCCC[C@H]3N)n3ncnc3c2C(N)=O)cc(OC)c1 |r| |
|---|
| Structure | 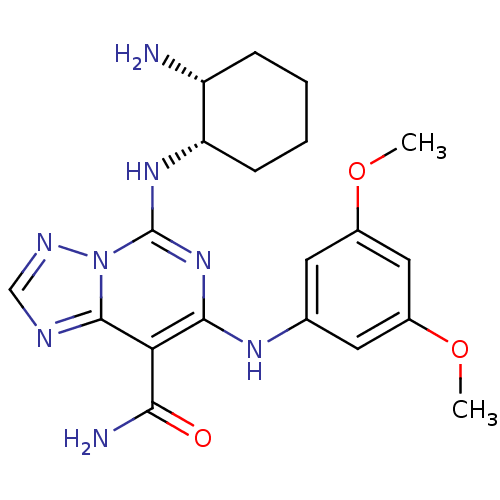
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Hirabayashi, A; Mukaiyama, H; Kobayashi, H; Shiohara, H; Nakayama, S; Ozawa, M; Miyazawa, K; Misawa, K; Ohnota, H; Isaji, M A novel Syk family kinase inhibitor: design, synthesis, and structure-activity relationship of 1,2,4-triazolo[4,3-c]pyrimidine and 1,2,4-triazolo[1,5-c]pyrimidine derivatives. Bioorg Med Chem16:7347-57 (2008) [PubMed] Article
Hirabayashi, A; Mukaiyama, H; Kobayashi, H; Shiohara, H; Nakayama, S; Ozawa, M; Miyazawa, K; Misawa, K; Ohnota, H; Isaji, M A novel Syk family kinase inhibitor: design, synthesis, and structure-activity relationship of 1,2,4-triazolo[4,3-c]pyrimidine and 1,2,4-triazolo[1,5-c]pyrimidine derivatives. Bioorg Med Chem16:7347-57 (2008) [PubMed] Article