| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Gamma-aminobutyric acid receptor subunit alpha-1 |
|---|
| Ligand | BDBM24183 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_514084 (CHEMBL979936) |
|---|
| EC50 | 10000±n/a nM |
|---|
| Citation |  Jansen, M; Rabe, H; Strehle, A; Dieler, S; Debus, F; Dannhardt, G; Akabas, MH; Lüddens, H Synthesis of GABAA receptor agonists and evaluation of their alpha-subunit selectivity and orientation in the GABA binding site. J Med Chem51:4430-48 (2008) [PubMed] Article Jansen, M; Rabe, H; Strehle, A; Dieler, S; Debus, F; Dannhardt, G; Akabas, MH; Lüddens, H Synthesis of GABAA receptor agonists and evaluation of their alpha-subunit selectivity and orientation in the GABA binding site. J Med Chem51:4430-48 (2008) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Gamma-aminobutyric acid receptor subunit alpha-1 |
|---|
| Name: | Gamma-aminobutyric acid receptor subunit alpha-1 |
|---|
| Synonyms: | Benzodiazepine central | Benzodiazepine receptors | GABA A Benzodiazepine | GABA A Benzodiazepine Type I | GABA A Benzodiazepine Type II | GABA A Benzodiazepine Type IIL | GABA A Benzodiazepine Type IIM | GABA A Benzodiazepine omega1 | GABA A Benzodiazepine omega2 | GABA A Benzodiazepine omega5 | GABA A alpha1 | GABA A anti-Alpha1 | GABA receptor alpha-1 subunit | GABA, Chloride, TBOB | GABA-PICROTOXIN | GBRA1_RAT | Gabra-1 | Gabra1 | TBPS |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 51770.21 |
|---|
| Organism: | Rattus norvegicus (Rat) |
|---|
| Description: | P62813 |
|---|
| Residue: | 455 |
|---|
| Sequence: | MKKSRGLSDYLWAWTLILSTLSGRSYGQPSQDELKDNTTVFTRILDRLLDGYDNRLRPGL
GERVTEVKTDIFVTSFGPVSDHDMEYTIDVFFRQSWKDERLKFKGPMTVLRLNNLMASKI
WTPDTFFHNGKKSVAHNMTMPNKLLRITEDGTLLYTMRLTVRAECPMHLEDFPMDAHACP
LKFGSYAYTRAEVVYEWTREPARSVVVAEDGSRLNQYDLLGQTVDSGIVQSSTGEYVVMT
THFHLKRKIGYFVIQTYLPCIMTVILSQVSFWLNRESVPARTVFGVTTVLTMTTLSISAR
NSLPKVAYATAMDWFIAVCYAFVFSALIEFATVNYFTKRGYAWDGKSVVPEKPKKVKDPL
IKKNNTYAPTATSYTPNLARGDPGLATIAKSATIEPKEVKPETKPPEPKKTFNSVSKIDR
LSRIAFPLLFGIFNLVYWATYLNREPQLKAPTPHQ
|
|
|
|---|
| BDBM24183 |
|---|
| n/a |
|---|
| Name | BDBM24183 |
|---|
| Synonyms: | 4-amino-n-[2,3-3H]butyric acid | 4-aminobutanoic acid | Aminalon | CHEMBL96 | GABA | Gamma-Aminobutyric acid | [3H]GABA | cid_119 |
|---|
| Type | Radiolabeled ligand |
|---|
| Emp. Form. | C4H9NO2 |
|---|
| Mol. Mass. | 103.1198 |
|---|
| SMILES | NCCCC(O)=O |
|---|
| Structure | 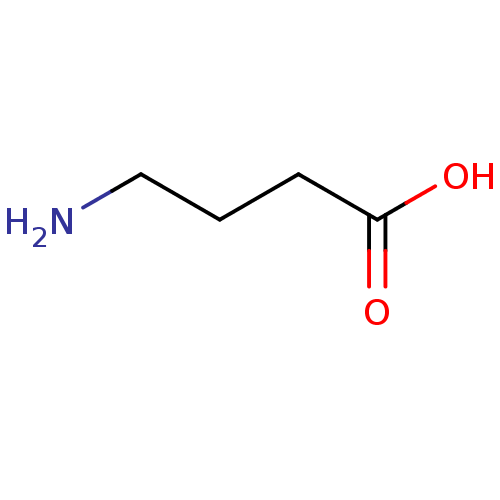
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Jansen, M; Rabe, H; Strehle, A; Dieler, S; Debus, F; Dannhardt, G; Akabas, MH; Lüddens, H Synthesis of GABAA receptor agonists and evaluation of their alpha-subunit selectivity and orientation in the GABA binding site. J Med Chem51:4430-48 (2008) [PubMed] Article
Jansen, M; Rabe, H; Strehle, A; Dieler, S; Debus, F; Dannhardt, G; Akabas, MH; Lüddens, H Synthesis of GABAA receptor agonists and evaluation of their alpha-subunit selectivity and orientation in the GABA binding site. J Med Chem51:4430-48 (2008) [PubMed] Article