| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Aryl hydrocarbon receptor |
|---|
| Ligand | BDBM50240990 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_510004 (CHEMBL1005597) |
|---|
| Ki | 6.8±n/a nM |
|---|
| Citation |  Aiello, S; Wells, G; Stone, EL; Kadri, H; Bazzi, R; Bell, DR; Stevens, MF; Matthews, CS; Bradshaw, TD; Westwell, AD Synthesis and biological properties of benzothiazole, benzoxazole, and chromen-4-one analogues of the potent antitumor agent 2-(3,4-dimethoxyphenyl)-5-fluorobenzothiazole (PMX 610, NSC 721648). J Med Chem51:5135-9 (2008) [PubMed] Article Aiello, S; Wells, G; Stone, EL; Kadri, H; Bazzi, R; Bell, DR; Stevens, MF; Matthews, CS; Bradshaw, TD; Westwell, AD Synthesis and biological properties of benzothiazole, benzoxazole, and chromen-4-one analogues of the potent antitumor agent 2-(3,4-dimethoxyphenyl)-5-fluorobenzothiazole (PMX 610, NSC 721648). J Med Chem51:5135-9 (2008) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Aryl hydrocarbon receptor |
|---|
| Name: | Aryl hydrocarbon receptor |
|---|
| Synonyms: | AHR_RAT | Ahr |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 96224.95 |
|---|
| Organism: | Rattus norvegicus |
|---|
| Description: | ChEMBL_510004 |
|---|
| Residue: | 853 |
|---|
| Sequence: | MSSGANITYASRKRRKPVQKTVKPVPAEGIKSNPSKRHRDRLNTELDRLASLLPFPQDVI
NKLDKLSVLRLSVSYLRAKSFFDVALKSTPADRSRGQDQCRAQVRDWQDLQEGEFLLQAL
NGFVLVVTADALVFYASSTIQDYLGFQQSDVIHQSVYELIHTEDRAEFQRQLHWALNPSQ
CTDSAQGVDETHGLPQPAVYYTPDQLPPENTAFMERCFRCRLRCLLDNSSGFLAMNFQGR
LKYLHGQNKKGKDGALLPPQLALFAIATPLQPPSILEIRTKNFIFRTKHKLDFTPIGCDA
KGQLILGYTEVELCNKGSGYQFIHAADMLHCAESHIRMIKTGESGMTVFRLLAKHSRWRW
VQSNARLIYRNGRPDYIIATQRPLTDEEGREHLQKRSMTLPFMFATGEAVLYEISSPFSP
IMDPLPIRTKSNTSRKDWAPQSTPSKDSFHPNSLMSALIQQDESIYLCPPSSPAPLDSHF
LMDSMSECGSWQGSFAVASNEALLKHEEIRHTQDVNLTLSGGPSELFPDNKNNDLYSIMR
NLGIDFEDIRSMQNEEFFRTDSSGEVDFKDIDITDEILTYVQDSLNNSTLLNSACQQQPV
SQHLSCMLQERLQLEQQQQLQQQHPTQTLEPQRQLCQVEVPQHELGQKTKHMQVNGMFAS
WNPAPPVSFSCPQQERKHYSLFSGLQGTAQEFPYKSEVDSMPYTQNFAPCNQSLLPEHSK
GTQLDFPGRDFERSLHPNASNLEDFVSCLQVPENQRHGINSQSAMVSPQAYYAGAMSMYQ
CQAGPQHTPVDQMQYSPEIPGSQAFLSKFQSPSILNEAYSADLSSIGHLQTAAHLPRLAE
AQPLPDITPSGFL
|
|
|
|---|
| BDBM50240990 |
|---|
| n/a |
|---|
| Name | BDBM50240990 |
|---|
| Synonyms: | 2-(3,4-dimethoxyphenyl)-5-fluorobenzothiazole | 5-fluoro-2-(3,4-dimethoxyphenyl)benzothiazole | CHEMBL373251 | NSC-721648 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C15H12FNO2S |
|---|
| Mol. Mass. | 289.325 |
|---|
| SMILES | COc1ccc(cc1OC)-c1nc2cc(F)ccc2s1 |
|---|
| Structure | 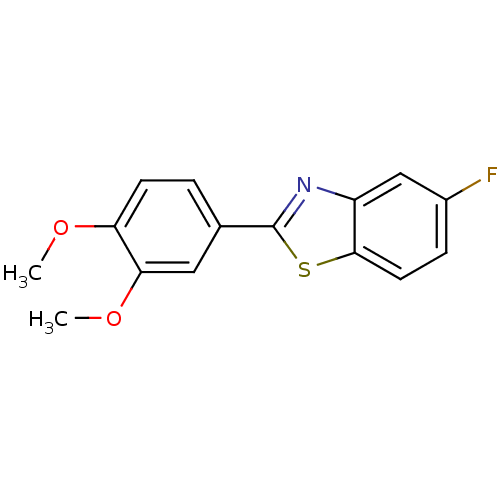
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Aiello, S; Wells, G; Stone, EL; Kadri, H; Bazzi, R; Bell, DR; Stevens, MF; Matthews, CS; Bradshaw, TD; Westwell, AD Synthesis and biological properties of benzothiazole, benzoxazole, and chromen-4-one analogues of the potent antitumor agent 2-(3,4-dimethoxyphenyl)-5-fluorobenzothiazole (PMX 610, NSC 721648). J Med Chem51:5135-9 (2008) [PubMed] Article
Aiello, S; Wells, G; Stone, EL; Kadri, H; Bazzi, R; Bell, DR; Stevens, MF; Matthews, CS; Bradshaw, TD; Westwell, AD Synthesis and biological properties of benzothiazole, benzoxazole, and chromen-4-one analogues of the potent antitumor agent 2-(3,4-dimethoxyphenyl)-5-fluorobenzothiazole (PMX 610, NSC 721648). J Med Chem51:5135-9 (2008) [PubMed] Article