| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cannabinoid receptor 1 |
|---|
| Ligand | BDBM50253805 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_510960 (CHEMBL1003147) |
|---|
| IC50 | 7±n/a nM |
|---|
| Citation |  Tseng, SL; Hung, MS; Chang, CP; Song, JS; Tai, CL; Chiu, HH; Hsieh, WP; Lin, Y; Chung, WL; Kuo, CW; Wu, CH; Chu, CM; Tung, YS; Chao, YS; Shia, KS Bioisosteric replacement of the pyrazole 5-aryl moiety of N-(piperidin-1-yl)-5-(4-chlorophenyl)-1-(2,4-dichlorophenyl)-4-methyl-1H-pyrazole-3-carboxamide (SR141716A). A novel series of alkynylthiophenes as potent and selective cannabinoid-1 receptor antagonists. J Med Chem51:5397-412 (2008) [PubMed] Article Tseng, SL; Hung, MS; Chang, CP; Song, JS; Tai, CL; Chiu, HH; Hsieh, WP; Lin, Y; Chung, WL; Kuo, CW; Wu, CH; Chu, CM; Tung, YS; Chao, YS; Shia, KS Bioisosteric replacement of the pyrazole 5-aryl moiety of N-(piperidin-1-yl)-5-(4-chlorophenyl)-1-(2,4-dichlorophenyl)-4-methyl-1H-pyrazole-3-carboxamide (SR141716A). A novel series of alkynylthiophenes as potent and selective cannabinoid-1 receptor antagonists. J Med Chem51:5397-412 (2008) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cannabinoid receptor 1 |
|---|
| Name: | Cannabinoid receptor 1 |
|---|
| Synonyms: | CANN6 | CANNABINOID CB1 | CB-R | CB1 | CNR | CNR1 | CNR1_HUMAN | Cannabinoid CB1 receptor | Cannabinoid receptor | Cannabinoid receptor 1 (CB1) | Cannabinoid receptor 1 (brain) |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 52868.96 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P21554 |
|---|
| Residue: | 472 |
|---|
| Sequence: | MKSILDGLADTTFRTITTDLLYVGSNDIQYEDIKGDMASKLGYFPQKFPLTSFRGSPFQE
KMTAGDNPQLVPADQVNITEFYNKSLSSFKENEENIQCGENFMDIECFMVLNPSQQLAIA
VLSLTLGTFTVLENLLVLCVILHSRSLRCRPSYHFIGSLAVADLLGSVIFVYSFIDFHVF
HRKDSRNVFLFKLGGVTASFTASVGSLFLTAIDRYISIHRPLAYKRIVTRPKAVVAFCLM
WTIAIVIAVLPLLGWNCEKLQSVCSDIFPHIDETYLMFWIGVTSVLLLFIVYAYMYILWK
AHSHAVRMIQRGTQKSIIIHTSEDGKVQVTRPDQARMDIRLAKTLVLILVVLIICWGPLL
AIMVYDVFGKMNKLIKTVFAFCSMLCLLNSTVNPIIYALRSKDLRHAFRSMFPSCEGTAQ
PLDNSMGDSDCLHKHANNAASVHRAAESCIKSTVKIAKVTMSVSTDTSAEAL
|
|
|
|---|
| BDBM50253805 |
|---|
| n/a |
|---|
| Name | BDBM50253805 |
|---|
| Synonyms: | CHEMBL460132 | N-(Azepan-1-yl)-1-(2,4-dichlorophenyl)-4-methyl-5-(5-(pent-1-ynyl)thiophen-2-yl)-1H-pyrazole-3-carboxamide |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C26H28Cl2N4OS |
|---|
| Mol. Mass. | 515.498 |
|---|
| SMILES | CCCC#Cc1ccc(s1)-c1c(C)c(nn1-c1ccc(Cl)cc1Cl)C(=O)NN1CCCCCC1 |
|---|
| Structure | 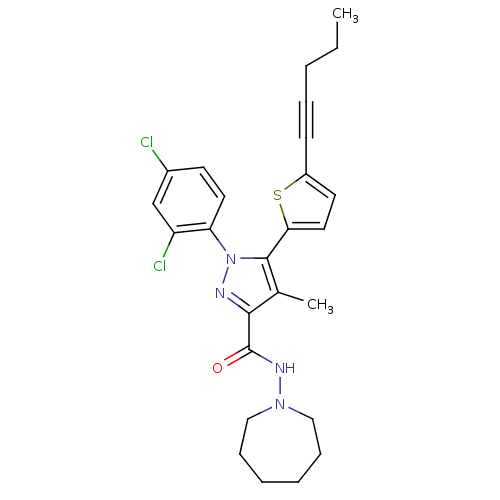
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Tseng, SL; Hung, MS; Chang, CP; Song, JS; Tai, CL; Chiu, HH; Hsieh, WP; Lin, Y; Chung, WL; Kuo, CW; Wu, CH; Chu, CM; Tung, YS; Chao, YS; Shia, KS Bioisosteric replacement of the pyrazole 5-aryl moiety of N-(piperidin-1-yl)-5-(4-chlorophenyl)-1-(2,4-dichlorophenyl)-4-methyl-1H-pyrazole-3-carboxamide (SR141716A). A novel series of alkynylthiophenes as potent and selective cannabinoid-1 receptor antagonists. J Med Chem51:5397-412 (2008) [PubMed] Article
Tseng, SL; Hung, MS; Chang, CP; Song, JS; Tai, CL; Chiu, HH; Hsieh, WP; Lin, Y; Chung, WL; Kuo, CW; Wu, CH; Chu, CM; Tung, YS; Chao, YS; Shia, KS Bioisosteric replacement of the pyrazole 5-aryl moiety of N-(piperidin-1-yl)-5-(4-chlorophenyl)-1-(2,4-dichlorophenyl)-4-methyl-1H-pyrazole-3-carboxamide (SR141716A). A novel series of alkynylthiophenes as potent and selective cannabinoid-1 receptor antagonists. J Med Chem51:5397-412 (2008) [PubMed] Article