| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Melanocortin receptor 4 |
|---|
| Ligand | BDBM50189008 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_538608 (CHEMBL1027398) |
|---|
| Ki | 11±n/a nM |
|---|
| Citation |  Tian, X; Switzer, AG; Derose, SA; Mishra, RK; Solinsky, MG; Mumin, RN; Ebetino, FH; Jayasinghe, LR; Webster, ME; Colson, AO; Crossdoersen, D; Pinney, BB; Farmer, JA; Dowty, ME; Obringer, CM; Cruze, CA; Burklow, ML; Suchanek, PM; Dong, L; Dirr, MK; Sheldon, RJ; Wos, JA Discovery of orally bioavailable 1,3,4-trisubstituted 2-oxopiperazine-based melanocortin-4 receptor agonists as potential antiobesity agents. J Med Chem51:6055-66 (2008) [PubMed] Article Tian, X; Switzer, AG; Derose, SA; Mishra, RK; Solinsky, MG; Mumin, RN; Ebetino, FH; Jayasinghe, LR; Webster, ME; Colson, AO; Crossdoersen, D; Pinney, BB; Farmer, JA; Dowty, ME; Obringer, CM; Cruze, CA; Burklow, ML; Suchanek, PM; Dong, L; Dirr, MK; Sheldon, RJ; Wos, JA Discovery of orally bioavailable 1,3,4-trisubstituted 2-oxopiperazine-based melanocortin-4 receptor agonists as potential antiobesity agents. J Med Chem51:6055-66 (2008) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Melanocortin receptor 4 |
|---|
| Name: | Melanocortin receptor 4 |
|---|
| Synonyms: | MC4-R | MC4R | MC4R_HUMAN | Melanocortin MC4 | Melanocortin receptor 4 (MC-4) | Melanocortin receptor 4 (MC4-R) | Melanocortin receptor 4 (MC4R) |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 36949.50 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P32245 |
|---|
| Residue: | 332 |
|---|
| Sequence: | MVNSTHRGMHTSLHLWNRSSYRLHSNASESLGKGYSDGGCYEQLFVSPEVFVTLGVISLL
ENILVIVAIAKNKNLHSPMYFFICSLAVADMLVSVSNGSETIVITLLNSTDTDAQSFTVN
IDNVIDSVICSSLLASICSLLSIAVDRYFTIFYALQYHNIMTVKRVGIIISCIWAACTVS
GILFIIYSDSSAVIICLITMFFTMLALMASLYVHMFLMARLHIKRIAVLPGTGAIRQGAN
MKGAITLTILIGVFVVCWAPFFLHLIFYISCPQNPYCVCFMSHFNLYLILIMCNSIIDPL
IYALRSQELRKTFKEIICCYPLGGLCDLSSRY
|
|
|
|---|
| BDBM50189008 |
|---|
| n/a |
|---|
| Name | BDBM50189008 |
|---|
| Synonyms: | (S)-2-((S)-4-((R)-2-amino-3-(4-fluorophenyl)propanoyl)-3-(2-guanidinoethyl)-2-oxopiperazin-1-yl)-N-methyl-3-(naphthalen-2-yl)propanamide | (S)-2-[(S)-4-[(R)-2-Amino-3-(4-fluoro-phenyl)-propionyl]-3-(2-guanidino-ethyl)-2-oxo-piperazin-1-yl]-N-methyl-3-naphthalen-2-yl-propionamide | CHEMBL213566 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C30H36FN7O3 |
|---|
| Mol. Mass. | 561.6503 |
|---|
| SMILES | [#6]-[#7]-[#6](=O)-[#6@H](-[#6]-c1ccc2ccccc2c1)-[#7]-1-[#6]-[#6]-[#7](-[#6@@H](-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#6]-1=O)-[#6](=O)-[#6@H](-[#7])-[#6]-c1ccc(F)cc1 |r| |
|---|
| Structure | 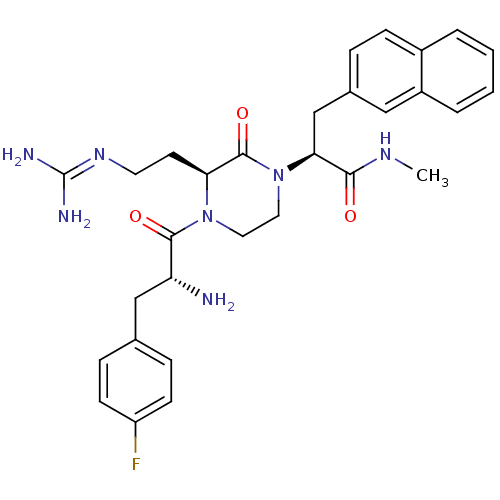
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Tian, X; Switzer, AG; Derose, SA; Mishra, RK; Solinsky, MG; Mumin, RN; Ebetino, FH; Jayasinghe, LR; Webster, ME; Colson, AO; Crossdoersen, D; Pinney, BB; Farmer, JA; Dowty, ME; Obringer, CM; Cruze, CA; Burklow, ML; Suchanek, PM; Dong, L; Dirr, MK; Sheldon, RJ; Wos, JA Discovery of orally bioavailable 1,3,4-trisubstituted 2-oxopiperazine-based melanocortin-4 receptor agonists as potential antiobesity agents. J Med Chem51:6055-66 (2008) [PubMed] Article
Tian, X; Switzer, AG; Derose, SA; Mishra, RK; Solinsky, MG; Mumin, RN; Ebetino, FH; Jayasinghe, LR; Webster, ME; Colson, AO; Crossdoersen, D; Pinney, BB; Farmer, JA; Dowty, ME; Obringer, CM; Cruze, CA; Burklow, ML; Suchanek, PM; Dong, L; Dirr, MK; Sheldon, RJ; Wos, JA Discovery of orally bioavailable 1,3,4-trisubstituted 2-oxopiperazine-based melanocortin-4 receptor agonists as potential antiobesity agents. J Med Chem51:6055-66 (2008) [PubMed] Article