| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Tyrosine-protein kinase ITK/TSK |
|---|
| Ligand | BDBM26827 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_537167 (CHEMBL992543) |
|---|
| IC50 | 10±n/a nM |
|---|
| Citation |  Moriarty, KJ; Winters, M; Qiao, L; Ryan, D; DesJarlis, R; Robinson, D; Cook, BN; Kashem, MA; Kaplita, PV; Liu, LH; Farrell, TM; Khine, HH; King, J; Pullen, SS; Roth, GP; Magolda, R; Takahashi, H Itk kinase inhibitors: initial efforts to improve the metabolical stability and the cell activity of the benzimidazole lead. Bioorg Med Chem Lett18:5537-40 (2008) [PubMed] Article Moriarty, KJ; Winters, M; Qiao, L; Ryan, D; DesJarlis, R; Robinson, D; Cook, BN; Kashem, MA; Kaplita, PV; Liu, LH; Farrell, TM; Khine, HH; King, J; Pullen, SS; Roth, GP; Magolda, R; Takahashi, H Itk kinase inhibitors: initial efforts to improve the metabolical stability and the cell activity of the benzimidazole lead. Bioorg Med Chem Lett18:5537-40 (2008) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Tyrosine-protein kinase ITK/TSK |
|---|
| Name: | Tyrosine-protein kinase ITK/TSK |
|---|
| Synonyms: | EMT | ITK | ITK_HUMAN | Kinase EMT | LYK | T-cell-specific kinase | Tyrosine-protein kinase ITK | Tyrosine-protein kinase ITK (ITK) | Tyrosine-protein kinase Lyk |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 71839.20 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Q08881 |
|---|
| Residue: | 620 |
|---|
| Sequence: | MNNFILLEEQLIKKSQQKRRTSPSNFKVRFFVLTKASLAYFEDRHGKKRTLKGSIELSRI
KCVEIVKSDISIPCHYKYPFQVVHDNYLLYVFAPDRESRQRWVLALKEETRNNNSLVPKY
HPNFWMDGKWRCCSQLEKLATGCAQYDPTKNASKKPLPPTPEDNRRPLWEPEETVVIALY
DYQTNDPQELALRRNEEYCLLDSSEIHWWRVQDRNGHEGYVPSSYLVEKSPNNLETYEWY
NKSISRDKAEKLLLDTGKEGAFMVRDSRTAGTYTVSVFTKAVVSENNPCIKHYHIKETND
NPKRYYVAEKYVFDSIPLLINYHQHNGGGLVTRLRYPVCFGRQKAPVTAGLRYGKWVIDP
SELTFVQEIGSGQFGLVHLGYWLNKDKVAIKTIREGAMSEEDFIEEAEVMMKLSHPKLVQ
LYGVCLEQAPICLVFEFMEHGCLSDYLRTQRGLFAAETLLGMCLDVCEGMAYLEEACVIH
RDLAARNCLVGENQVIKVSDFGMTRFVLDDQYTSSTGTKFPVKWASPEVFSFSRYSSKSD
VWSFGVLMWEVFSEGKIPYENRSNSEVVEDISTGFRLYKPRLASTHVYQIMNHCWKERPE
DRPAFSRLLRQLAEIAESGL
|
|
|
|---|
| BDBM26827 |
|---|
| n/a |
|---|
| Name | BDBM26827 |
|---|
| Synonyms: | CHEMBL458796 | N-[(2E)-5-[benzene(methyl)amido]-1-(2-carbamoylethyl)-2,3-dihydro-1H-1,3-benzodiazol-2-ylidene]-1,2-oxazole-5-carboxamide | acylated 2-aminobenzimidazole, 21 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C22H20N6O4 |
|---|
| Mol. Mass. | 432.432 |
|---|
| SMILES | CN(C(=O)c1ccccc1)c1ccc2n(CCC(N)=O)c(NC(=O)c3ccno3)nc2c1 |
|---|
| Structure | 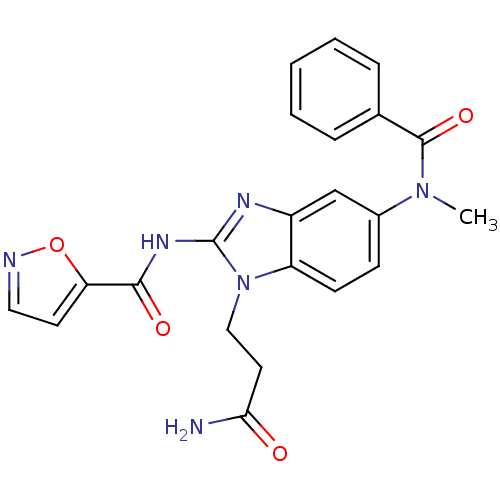
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Moriarty, KJ; Winters, M; Qiao, L; Ryan, D; DesJarlis, R; Robinson, D; Cook, BN; Kashem, MA; Kaplita, PV; Liu, LH; Farrell, TM; Khine, HH; King, J; Pullen, SS; Roth, GP; Magolda, R; Takahashi, H Itk kinase inhibitors: initial efforts to improve the metabolical stability and the cell activity of the benzimidazole lead. Bioorg Med Chem Lett18:5537-40 (2008) [PubMed] Article
Moriarty, KJ; Winters, M; Qiao, L; Ryan, D; DesJarlis, R; Robinson, D; Cook, BN; Kashem, MA; Kaplita, PV; Liu, LH; Farrell, TM; Khine, HH; King, J; Pullen, SS; Roth, GP; Magolda, R; Takahashi, H Itk kinase inhibitors: initial efforts to improve the metabolical stability and the cell activity of the benzimidazole lead. Bioorg Med Chem Lett18:5537-40 (2008) [PubMed] Article