

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Reaction Details | |||
|---|---|---|---|
 | Report a problem with these data | ||
| Target | Cytochrome P450 2D6 | ||
| Ligand | BDBM50198075 | ||
| Substrate/Competitor | n/a | ||
| Meas. Tech. | ChEMBL_515428 (CHEMBL1028887) | ||
| Ki | 450±n/a nM | ||
| Citation |  Ponte-Sucre, A; Gulder, T; Wegehaupt, A; Albert, C; Rikanovic, C; Schaeflein, L; Frank, A; Schultheis, M; Unger, M; Holzgrabe, U; Bringmann, G; Moll, H Structure-activity relationship and studies on the molecular mechanism of leishmanicidal N,C-coupled arylisoquinolinium salts. J Med Chem52:626-36 (2009) [PubMed] Article Ponte-Sucre, A; Gulder, T; Wegehaupt, A; Albert, C; Rikanovic, C; Schaeflein, L; Frank, A; Schultheis, M; Unger, M; Holzgrabe, U; Bringmann, G; Moll, H Structure-activity relationship and studies on the molecular mechanism of leishmanicidal N,C-coupled arylisoquinolinium salts. J Med Chem52:626-36 (2009) [PubMed] Article | ||
| More Info.: | Get all data from this article, Assay Method | ||
| Cytochrome P450 2D6 | |||
| Name: | Cytochrome P450 2D6 | ||
| Synonyms: | CP2D6_HUMAN | CYP2D6 | CYP2DL1 | CYPIID6 | Cytochrome P450 2D6 (CYP2D6) | Debrisoquine 4-hydroxylase | P450-DB1 | ||
| Type: | Protein | ||
| Mol. Mass.: | 55774.82 | ||
| Organism: | Homo sapiens (Human) | ||
| Description: | P10635 | ||
| Residue: | 497 | ||
| Sequence: |
| ||
| BDBM50198075 | |||
| n/a | |||
| Name | BDBM50198075 | ||
| Synonyms: | 6,8-dimethoxy-1,3-dimethyl-2-(naphthalen-1-yl)isoquinolinium perchlorate | CHEMBL375355 | N,1'-naphthyl-6,8-dimethoxy-1,3-dimethylisoquinolinium perchlorate | ||
| Type | Small organic molecule | ||
| Emp. Form. | C23H22NO2 | ||
| Mol. Mass. | 344.4257 | ||
| SMILES | COc1cc(OC)c2c(C)[n+](c(C)cc2c1)-c1cccc2ccccc12 |(-7.05,6.23,;-7.05,4.69,;-5.73,3.91,;-5.73,2.37,;-4.39,1.6,;-4.39,.06,;-5.72,-.72,;-3.06,2.37,;-1.72,1.61,;-1.72,.07,;-.38,2.38,;-.39,3.93,;.94,4.71,;-1.73,4.7,;-3.06,3.92,;-4.4,4.69,;.96,1.62,;2.28,2.4,;3.61,1.64,;3.62,.09,;2.29,-.69,;2.3,-2.21,;.97,-2.98,;-.36,-2.21,;-.37,-.69,;.96,.09,)| | ||
| Structure | 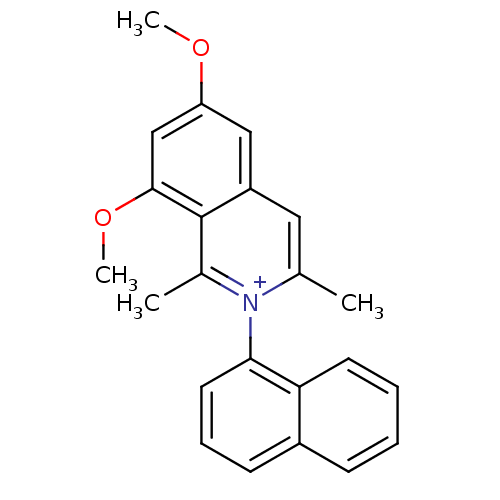
| ||