

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Reaction Details | |||
|---|---|---|---|
 | Report a problem with these data | ||
| Target | Kappa-type opioid receptor | ||
| Ligand | BDBM50159165 | ||
| Substrate/Competitor | n/a | ||
| Meas. Tech. | ChEMBL_517097 (CHEMBL988702) | ||
| EC50 | 2.1±n/a nM | ||
| Citation |  Béguin, C; Duncan, KK; Munro, TA; Ho, DM; Xu, W; Liu-Chen, LY; Carlezon, WA; Cohen, BM Modification of the furan ring of salvinorin A: identification of a selective partial agonist at the kappa opioid receptor. Bioorg Med Chem17:1370-80 (2009) [PubMed] Article Béguin, C; Duncan, KK; Munro, TA; Ho, DM; Xu, W; Liu-Chen, LY; Carlezon, WA; Cohen, BM Modification of the furan ring of salvinorin A: identification of a selective partial agonist at the kappa opioid receptor. Bioorg Med Chem17:1370-80 (2009) [PubMed] Article | ||
| More Info.: | Get all data from this article, Assay Method | ||
| Kappa-type opioid receptor | |||
| Name: | Kappa-type opioid receptor | ||
| Synonyms: | K-OR-1 | KOR-1 | Kappa-opioid receptor (KOR) | Kappa-type opioid receptor (KOPR) | Kappa-type opioid receptor (KOR) | Kappa-type opioid receptor (Kappa) | OPIATE Kappa | OPRK | OPRK1 | OPRK_HUMAN | kappa opioid receptor (KOR) | ||
| Type: | G Protein-Coupled Receptor (GPCR) | ||
| Mol. Mass.: | 42648.76 | ||
| Organism: | Homo sapiens (Human) | ||
| Description: | P41145 | ||
| Residue: | 380 | ||
| Sequence: |
| ||
| BDBM50159165 | |||
| n/a | |||
| Name | BDBM50159165 | ||
| Synonyms: | (2S,4aR,6aR,7R,9S,10aS,10bR)-9-(acetyloxy)-2-(furan-3-yl)dodecahydro-6a,10b-dimethyl-4,10-dioxo-2H-naphtho-[2,1-c]pyran-7-carboxylic acid methyl ester | (2S,4aR,6aR,7R,9S,10aS,10bR)-methyl 9-(acetylthio)-2-(furan-3-yl)-6a,10b-dimethyl-4,10-dioxo-dodecahydro-1H-benzo[f]isochromene-7-carboxylate | (2S,4aR,6aR,7R,9S,10aS,10bR)-methyl 9-acetoxy-2-(furan-3-yl)-6a,10b-dimethyl-4,10-dioxo-dodecahydro-1H-benzo[f]isochromene-7-carboxylate | (3S,4aR,4bS,6S,8R,8aR,10aR)-6-Acetoxy-3-furan-3-yl-4a,8a-dimethyl-1,5-dioxo-dodecahydro-2-oxa-phenanthrene-8-carboxylic acid methyl ester | 2S,4aR,6a(R,7R,9S,10aS,10bR)-9-(acetyloxy)-2-(furan-3-yl)dodecahydro-6a,10b-dimethyl-4,10-dioxo-2H-naphtho[2,1-c]pyran-7-carboxylic acid methyl ester | CHEMBL445332 | Salvinorin A | Salvonorin A | salvinorin-A | ||
| Type | Small organic molecule | ||
| Emp. Form. | C23H28O8 | ||
| Mol. Mass. | 432.4636 | ||
| SMILES | COC(=O)[C@@H]1C[C@H](OC(C)=O)C(=O)[C@H]2[C@@]1(C)CC[C@H]1C(=O)O[C@@H](C[C@]21C)c1ccoc1 |r| | ||
| Structure | 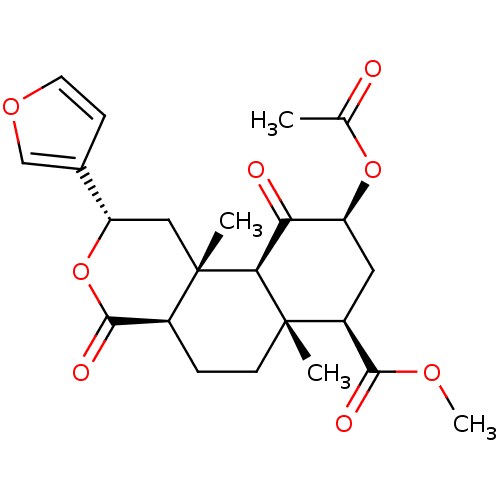
| ||