| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Coagulation factor X |
|---|
| Ligand | BDBM50266892 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_563985 (CHEMBL956028) |
|---|
| IC50 | 0.5±n/a nM |
|---|
| Citation |  Van Huis, CA; Casimiro-Garcia, A; Bigge, CF; Cody, WL; Dudley, DA; Filipski, KJ; Heemstra, RJ; Kohrt, JT; Leadley, RJ; Narasimhan, LS; McClanahan, T; Mochalkin, I; Pamment, M; Peterson, JT; Sahasrabudhe, V; Schaum, RP; Edmunds, JJ Exploration of 4,4-disubstituted pyrrolidine-1,2-dicarboxamides as potent, orally active Factor Xa inhibitors with extended duration of action. Bioorg Med Chem17:2501-11 (2009) [PubMed] Article Van Huis, CA; Casimiro-Garcia, A; Bigge, CF; Cody, WL; Dudley, DA; Filipski, KJ; Heemstra, RJ; Kohrt, JT; Leadley, RJ; Narasimhan, LS; McClanahan, T; Mochalkin, I; Pamment, M; Peterson, JT; Sahasrabudhe, V; Schaum, RP; Edmunds, JJ Exploration of 4,4-disubstituted pyrrolidine-1,2-dicarboxamides as potent, orally active Factor Xa inhibitors with extended duration of action. Bioorg Med Chem17:2501-11 (2009) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Coagulation factor X |
|---|
| Name: | Coagulation factor X |
|---|
| Synonyms: | Activated coagulation factor X (FXa) | Activated factor Xa heavy chain | Coagulation factor X precursor | Coagulation factor Xa | F10 | FA10_HUMAN | Factor X heavy chain | Factor X light chain | Factor Xa | Stuart factor | Stuart-Prower factor |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 54726.60 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | n/a |
|---|
| Residue: | 488 |
|---|
| Sequence: | MGRPLHLVLLSASLAGLLLLGESLFIRREQANNILARVTRANSFLEEMKKGHLERECMEE
TCSYEEAREVFEDSDKTNEFWNKYKDGDQCETSPCQNQGKCKDGLGEYTCTCLEGFEGKN
CELFTRKLCSLDNGDCDQFCHEEQNSVVCSCARGYTLADNGKACIPTGPYPCGKQTLERR
KRSVAQATSSSGEAPDSITWKPYDAADLDPTENPFDLLDFNQTQPERGDNNLTRIVGGQE
CKDGECPWQALLINEENEGFCGGTILSEFYILTAAHCLYQAKRFKVRVGDRNTEQEEGGE
AVHEVEVVIKHNRFTKETYDFDIAVLRLKTPITFRMNVAPACLPERDWAESTLMTQKTGI
VSGFGRTHEKGRQSTRLKMLEVPYVDRNSCKLSSSFIITQNMFCAGYDTKQEDACQGDSG
GPHVTRFKDTYFVTGIVSWGEGCARKGKYGIYTKVTAFLKWIDRSMKTRGLPKAKSHAPE
VITSSPLK
|
|
|
|---|
| BDBM50266892 |
|---|
| n/a |
|---|
| Name | BDBM50266892 |
|---|
| Synonyms: | (2R,4R)-N1-(4-Chlorophenyl)-4-ethyl-N2-(2-fluoro-4-(3-methyl-2-oxopyridin-1(2H)-yl)phenyl)-4-hydroxypyrrolidine-1,2-dicarboxamide | CHEMBL515156 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C26H26ClFN4O4 |
|---|
| Mol. Mass. | 512.96 |
|---|
| SMILES | CC[C@@]1(O)C[C@@H](N(C1)C(=O)Nc1ccc(Cl)cc1)C(=O)Nc1ccc(cc1F)-n1cccc(C)c1=O |r| |
|---|
| Structure | 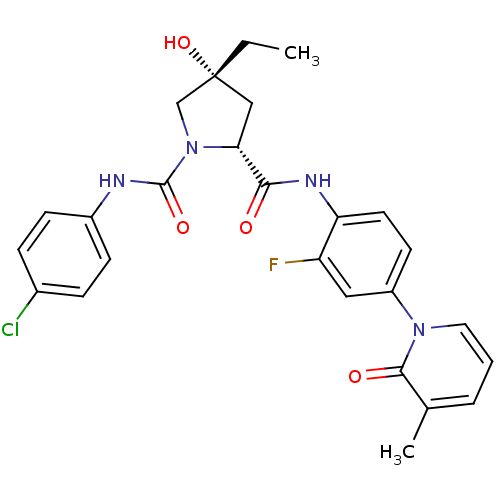
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Van Huis, CA; Casimiro-Garcia, A; Bigge, CF; Cody, WL; Dudley, DA; Filipski, KJ; Heemstra, RJ; Kohrt, JT; Leadley, RJ; Narasimhan, LS; McClanahan, T; Mochalkin, I; Pamment, M; Peterson, JT; Sahasrabudhe, V; Schaum, RP; Edmunds, JJ Exploration of 4,4-disubstituted pyrrolidine-1,2-dicarboxamides as potent, orally active Factor Xa inhibitors with extended duration of action. Bioorg Med Chem17:2501-11 (2009) [PubMed] Article
Van Huis, CA; Casimiro-Garcia, A; Bigge, CF; Cody, WL; Dudley, DA; Filipski, KJ; Heemstra, RJ; Kohrt, JT; Leadley, RJ; Narasimhan, LS; McClanahan, T; Mochalkin, I; Pamment, M; Peterson, JT; Sahasrabudhe, V; Schaum, RP; Edmunds, JJ Exploration of 4,4-disubstituted pyrrolidine-1,2-dicarboxamides as potent, orally active Factor Xa inhibitors with extended duration of action. Bioorg Med Chem17:2501-11 (2009) [PubMed] Article