| Reaction Details |
|---|
 | Report a problem with these data |
| Target | 5-hydroxytryptamine receptor 2A |
|---|
| Ligand | BDBM50113332 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_563483 (CHEMBL961031) |
|---|
| Ki | 0.13±n/a nM |
|---|
| Citation |  Herth, MM; Kramer, V; Piel, M; Palner, M; Riss, PJ; Knudsen, GM; Rösch, F Synthesis and in vitro affinities of various MDL 100907 derivatives as potential 18F-radioligands for 5-HT2A receptor imaging with PET. Bioorg Med Chem17:2989-3002 (2009) [PubMed] Article Herth, MM; Kramer, V; Piel, M; Palner, M; Riss, PJ; Knudsen, GM; Rösch, F Synthesis and in vitro affinities of various MDL 100907 derivatives as potential 18F-radioligands for 5-HT2A receptor imaging with PET. Bioorg Med Chem17:2989-3002 (2009) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| 5-hydroxytryptamine receptor 2A |
|---|
| Name: | 5-hydroxytryptamine receptor 2A |
|---|
| Synonyms: | 5-HT-2A | 5-HT2 | 5-HT2A | 5-hydroxytryptamine receptor 2A (5-HT2A) | 5-hydroxytryptamine receptor 2A (5HT2A) | 5HT2A_RAT | Htr2 | Htr2a | Serotonin Receptor 2A |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 52852.05 |
|---|
| Organism: | Rattus norvegicus (rat) |
|---|
| Description: | Rat cortex membranes 5-HT2A receptors. |
|---|
| Residue: | 471 |
|---|
| Sequence: | MEILCEDNISLSSIPNSLMQLGDGPRLYHNDFNSRDANTSEASNWTIDAENRTNLSCEGY
LPPTCLSILHLQEKNWSALLTTVVIILTIAGNILVIMAVSLEKKLQNATNYFLMSLAIAD
MLLGFLVMPVSMLTILYGYRWPLPSKLCAIWIYLDVLFSTASIMHLCAISLDRYVAIQNP
IHHSRFNSRTKAFLKIIAVWTISVGISMPIPVFGLQDDSKVFKEGSCLLADDNFVLIGSF
VAFFIPLTIMVITYFLTIKSLQKEATLCVSDLSTRAKLASFSFLPQSSLSSEKLFQRSIH
REPGSYAGRRTMQSISNEQKACKVLGIVFFLFVVMWCPFFITNIMAVICKESCNENVIGA
LLNVFVWIGYLSSAVNPLVYTLFNKTYRSAFSRYIQCQYKENRKPLQLILVNTIPALAYK
SSQLQVGQKKNSQEDAEQTVDDCSMVTLGKQQSEENCTDNIETVNEKVSCV
|
|
|
|---|
| BDBM50113332 |
|---|
| n/a |
|---|
| Name | BDBM50113332 |
|---|
| Synonyms: | 3-(2-(4-(4-fluorobenzoyl)piperidin-1-yl)ethyl)-2-thioxo-2,3-dihydroquinazolin-4(1H)-one | 3-{2-[4-(4-Fluoro-benzoyl)-piperidin-1-yl]-ethyl}-2-thioxo-2,3-dihydro-1H-quinazolin-4-one | CHEMBL62919 | altanserin |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C22H22FN3O2S |
|---|
| Mol. Mass. | 411.492 |
|---|
| SMILES | Fc1ccc(cc1)C(=O)C1CCN(CCn2c(=S)[nH]c3ccccc3c2=O)CC1 |
|---|
| Structure | 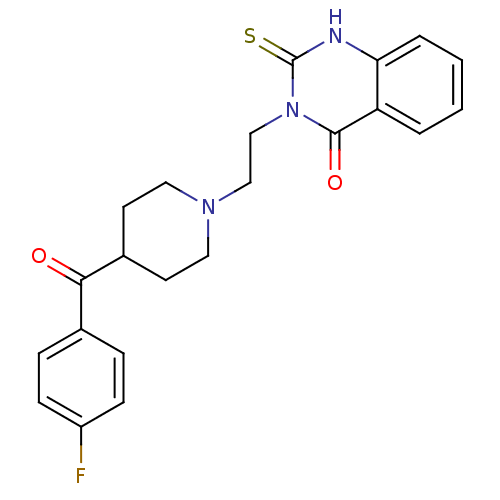
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Herth, MM; Kramer, V; Piel, M; Palner, M; Riss, PJ; Knudsen, GM; Rösch, F Synthesis and in vitro affinities of various MDL 100907 derivatives as potential 18F-radioligands for 5-HT2A receptor imaging with PET. Bioorg Med Chem17:2989-3002 (2009) [PubMed] Article
Herth, MM; Kramer, V; Piel, M; Palner, M; Riss, PJ; Knudsen, GM; Rösch, F Synthesis and in vitro affinities of various MDL 100907 derivatives as potential 18F-radioligands for 5-HT2A receptor imaging with PET. Bioorg Med Chem17:2989-3002 (2009) [PubMed] Article