| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Neuropeptide Y receptor type 5 |
|---|
| Ligand | BDBM50249737 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_498773 (CHEMBL1021965) |
|---|
| Ki | 2±n/a nM |
|---|
| Citation |  Sato, N; Ando, M; Ishikawa, S; Jitsuoka, M; Nagai, K; Takahashi, H; Sakuraba, A; Tsuge, H; Kitazawa, H; Iwaasa, H; Mashiko, S; Gomori, A; Moriya, R; Fujino, N; Ohe, T; Ishihara, A; Kanatani, A; Fukami, T Discovery of tetrasubstituted imidazolines as potent and selective neuropeptide Y Y5 receptor antagonists: reduced human ether-a-go-go related gene potassium channel binding affinity and potent antiobesity effect. J Med Chem52:3385-96 (2009) [PubMed] Article Sato, N; Ando, M; Ishikawa, S; Jitsuoka, M; Nagai, K; Takahashi, H; Sakuraba, A; Tsuge, H; Kitazawa, H; Iwaasa, H; Mashiko, S; Gomori, A; Moriya, R; Fujino, N; Ohe, T; Ishihara, A; Kanatani, A; Fukami, T Discovery of tetrasubstituted imidazolines as potent and selective neuropeptide Y Y5 receptor antagonists: reduced human ether-a-go-go related gene potassium channel binding affinity and potent antiobesity effect. J Med Chem52:3385-96 (2009) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Neuropeptide Y receptor type 5 |
|---|
| Name: | Neuropeptide Y receptor type 5 |
|---|
| Synonyms: | NPY-Y5 | NPY-Y5 receptor | NPY5-R | NPY5R | NPY5R_HUMAN | NPYR5 | NPYY5 | Neuropeptide Y receptor type 5 | Neuropeptide Y receptor type 5 ( NPY Y5) | Y5 receptor |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 50746.64 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Q15761 |
|---|
| Residue: | 445 |
|---|
| Sequence: | MDLELDEYYNKTLATENNTAATRNSDFPVWDDYKSSVDDLQYFLIGLYTFVSLLGFMGNL
LILMALMKKRNQKTTVNFLIGNLAFSDILVVLFCSPFTLTSVLLDQWMFGKVMCHIMPFL
QCVSVLVSTLILISIAIVRYHMIKHPISNNLTANHGYFLIATVWTLGFAICSPLPVFHSL
VELQETFGSALLSSRYLCVESWPSDSYRIAFTISLLLVQYILPLVCLTVSHTSVCRSISC
GLSNKENRLEENEMINLTLHPSKKSGPQVKLSGSHKWSYSFIKKHRRRYSKKTACVLPAP
ERPSQENHSRILPENFGSVRSQLSSSSKFIPGVPTCFEIKPEENSDVHELRVKRSVTRIK
KRSRSVFYRLTILILVFAVSWMPLHLFHVVTDFNDNLISNRHFKLVYCICHLLGMMSCCL
NPILYGFLNNGIKADLVSLIHCLHM
|
|
|
|---|
| BDBM50249737 |
|---|
| n/a |
|---|
| Name | BDBM50249737 |
|---|
| Synonyms: | 2-(4,4-bis(4-fluorophenyl)-4,5-dihydro-1H-imidazol-2-yl)isonicotinonitrile | CHEMBL522456 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C21H14F2N4 |
|---|
| Mol. Mass. | 360.3595 |
|---|
| SMILES | Fc1ccc(cc1)C1(CNC(=N1)c1cc(ccn1)C#N)c1ccc(F)cc1 |c:11| |
|---|
| Structure | 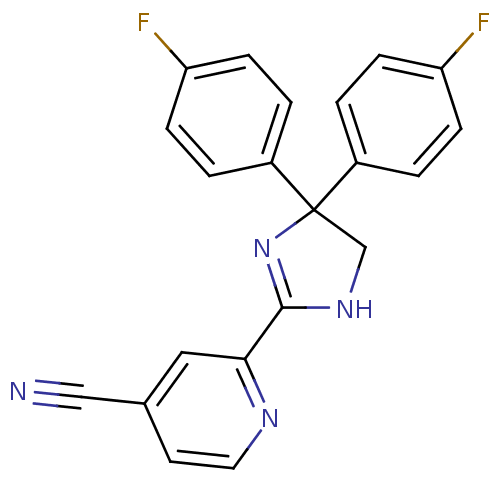
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Sato, N; Ando, M; Ishikawa, S; Jitsuoka, M; Nagai, K; Takahashi, H; Sakuraba, A; Tsuge, H; Kitazawa, H; Iwaasa, H; Mashiko, S; Gomori, A; Moriya, R; Fujino, N; Ohe, T; Ishihara, A; Kanatani, A; Fukami, T Discovery of tetrasubstituted imidazolines as potent and selective neuropeptide Y Y5 receptor antagonists: reduced human ether-a-go-go related gene potassium channel binding affinity and potent antiobesity effect. J Med Chem52:3385-96 (2009) [PubMed] Article
Sato, N; Ando, M; Ishikawa, S; Jitsuoka, M; Nagai, K; Takahashi, H; Sakuraba, A; Tsuge, H; Kitazawa, H; Iwaasa, H; Mashiko, S; Gomori, A; Moriya, R; Fujino, N; Ohe, T; Ishihara, A; Kanatani, A; Fukami, T Discovery of tetrasubstituted imidazolines as potent and selective neuropeptide Y Y5 receptor antagonists: reduced human ether-a-go-go related gene potassium channel binding affinity and potent antiobesity effect. J Med Chem52:3385-96 (2009) [PubMed] Article