| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cannabinoid receptor 1 |
|---|
| Ligand | BDBM50279204 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_545295 (CHEMBL1020741) |
|---|
| IC50 | 14.6±n/a nM |
|---|
| Citation |  Wu, CH; Hung, MS; Song, JS; Yeh, TK; Chou, MC; Chu, CM; Jan, JJ; Hsieh, MT; Tseng, SL; Chang, CP; Hsieh, WP; Lin, Y; Yeh, YN; Chung, WL; Kuo, CW; Lin, CY; Shy, HS; Chao, YS; Shia, KS Discovery of 2-[5-(4-chloro-phenyl)-1-(2,4-dichloro-phenyl)-4-ethyl-1H-pyrazol-3-yl]-1,5,5-trimethyl-1,5-dihydro-imidazol-4-thione (BPR-890) via an active metabolite. A novel, potent and selective cannabinoid-1 receptor inverse agonist with high antiobesity efficacy in DIO mice. J Med Chem52:4496-510 (2009) [PubMed] Article Wu, CH; Hung, MS; Song, JS; Yeh, TK; Chou, MC; Chu, CM; Jan, JJ; Hsieh, MT; Tseng, SL; Chang, CP; Hsieh, WP; Lin, Y; Yeh, YN; Chung, WL; Kuo, CW; Lin, CY; Shy, HS; Chao, YS; Shia, KS Discovery of 2-[5-(4-chloro-phenyl)-1-(2,4-dichloro-phenyl)-4-ethyl-1H-pyrazol-3-yl]-1,5,5-trimethyl-1,5-dihydro-imidazol-4-thione (BPR-890) via an active metabolite. A novel, potent and selective cannabinoid-1 receptor inverse agonist with high antiobesity efficacy in DIO mice. J Med Chem52:4496-510 (2009) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cannabinoid receptor 1 |
|---|
| Name: | Cannabinoid receptor 1 |
|---|
| Synonyms: | CANN6 | CANNABINOID CB1 | CB-R | CB1 | CNR | CNR1 | CNR1_HUMAN | Cannabinoid CB1 receptor | Cannabinoid receptor | Cannabinoid receptor 1 (CB1) | Cannabinoid receptor 1 (brain) |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 52868.96 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P21554 |
|---|
| Residue: | 472 |
|---|
| Sequence: | MKSILDGLADTTFRTITTDLLYVGSNDIQYEDIKGDMASKLGYFPQKFPLTSFRGSPFQE
KMTAGDNPQLVPADQVNITEFYNKSLSSFKENEENIQCGENFMDIECFMVLNPSQQLAIA
VLSLTLGTFTVLENLLVLCVILHSRSLRCRPSYHFIGSLAVADLLGSVIFVYSFIDFHVF
HRKDSRNVFLFKLGGVTASFTASVGSLFLTAIDRYISIHRPLAYKRIVTRPKAVVAFCLM
WTIAIVIAVLPLLGWNCEKLQSVCSDIFPHIDETYLMFWIGVTSVLLLFIVYAYMYILWK
AHSHAVRMIQRGTQKSIIIHTSEDGKVQVTRPDQARMDIRLAKTLVLILVVLIICWGPLL
AIMVYDVFGKMNKLIKTVFAFCSMLCLLNSTVNPIIYALRSKDLRHAFRSMFPSCEGTAQ
PLDNSMGDSDCLHKHANNAASVHRAAESCIKSTVKIAKVTMSVSTDTSAEAL
|
|
|
|---|
| BDBM50279204 |
|---|
| n/a |
|---|
| Name | BDBM50279204 |
|---|
| Synonyms: | 2-[5-(4-Chloro-phenyl)-1-(2,4-dichloro-phenyl)-4-ethyl-1H-pyrazol-3-yl]-1,3-diaza-spiro[4.4]non-1-en-4-one | CHEMBL497914 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C24H21Cl3N4O |
|---|
| Mol. Mass. | 487.809 |
|---|
| SMILES | CCc1c(nn(c1-c1ccc(Cl)cc1)-c1ccc(Cl)cc1Cl)C1=NC2(CCCC2)C(=O)N1 |t:25| |
|---|
| Structure | 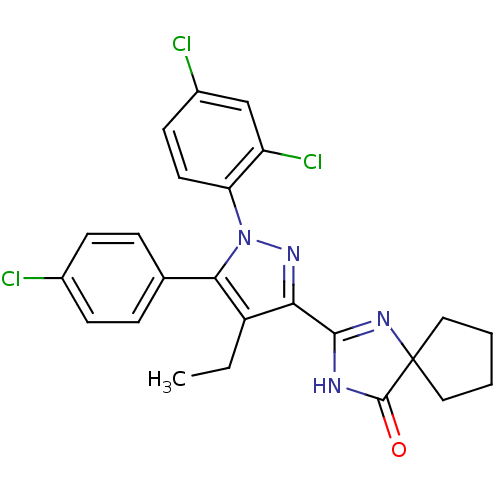
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Wu, CH; Hung, MS; Song, JS; Yeh, TK; Chou, MC; Chu, CM; Jan, JJ; Hsieh, MT; Tseng, SL; Chang, CP; Hsieh, WP; Lin, Y; Yeh, YN; Chung, WL; Kuo, CW; Lin, CY; Shy, HS; Chao, YS; Shia, KS Discovery of 2-[5-(4-chloro-phenyl)-1-(2,4-dichloro-phenyl)-4-ethyl-1H-pyrazol-3-yl]-1,5,5-trimethyl-1,5-dihydro-imidazol-4-thione (BPR-890) via an active metabolite. A novel, potent and selective cannabinoid-1 receptor inverse agonist with high antiobesity efficacy in DIO mice. J Med Chem52:4496-510 (2009) [PubMed] Article
Wu, CH; Hung, MS; Song, JS; Yeh, TK; Chou, MC; Chu, CM; Jan, JJ; Hsieh, MT; Tseng, SL; Chang, CP; Hsieh, WP; Lin, Y; Yeh, YN; Chung, WL; Kuo, CW; Lin, CY; Shy, HS; Chao, YS; Shia, KS Discovery of 2-[5-(4-chloro-phenyl)-1-(2,4-dichloro-phenyl)-4-ethyl-1H-pyrazol-3-yl]-1,5,5-trimethyl-1,5-dihydro-imidazol-4-thione (BPR-890) via an active metabolite. A novel, potent and selective cannabinoid-1 receptor inverse agonist with high antiobesity efficacy in DIO mice. J Med Chem52:4496-510 (2009) [PubMed] Article