| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 1A2 |
|---|
| Ligand | BDBM50293855 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_572590 (CHEMBL1025206) |
|---|
| IC50 | >100000±n/a nM |
|---|
| Citation |  Satoh, A; Sagara, T; Sakoh, H; Hashimoto, M; Nakashima, H; Kato, T; Goto, Y; Mizutani, S; Azuma-Kanoh, T; Tani, T; Okuda, S; Okamoto, O; Ozaki, S; Iwasawa, Y; Ohta, H; Kawamoto, H Identification of an orally active opioid receptor-like 1 (ORL1) receptor antagonist 4-{3-[(2R)-2,3-dihydroxypropyl]-2-oxo-2,3-dihydro-1H-benzimidazol-1-yl}-1-[(1S,3S,4R)-spiro[bicyclo[2.2.1]heptane-2,1'-cyclopropan]-3-ylmethyl]piperidine as clinical candidate. J Med Chem52:4091-4 (2009) [PubMed] Article Satoh, A; Sagara, T; Sakoh, H; Hashimoto, M; Nakashima, H; Kato, T; Goto, Y; Mizutani, S; Azuma-Kanoh, T; Tani, T; Okuda, S; Okamoto, O; Ozaki, S; Iwasawa, Y; Ohta, H; Kawamoto, H Identification of an orally active opioid receptor-like 1 (ORL1) receptor antagonist 4-{3-[(2R)-2,3-dihydroxypropyl]-2-oxo-2,3-dihydro-1H-benzimidazol-1-yl}-1-[(1S,3S,4R)-spiro[bicyclo[2.2.1]heptane-2,1'-cyclopropan]-3-ylmethyl]piperidine as clinical candidate. J Med Chem52:4091-4 (2009) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 1A2 |
|---|
| Name: | Cytochrome P450 1A2 |
|---|
| Synonyms: | CP1A2_HUMAN | CYP1A2 | CYPIA2 | Cholesterol 25-hydroxylase | Cytochrome P(3)450 | Cytochrome P450 1A | Cytochrome P450 1A2 (CYP1A2) | Cytochrome P450 4 | Cytochrome P450-P3 |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 58423.38 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P05177 |
|---|
| Residue: | 516 |
|---|
| Sequence: | MALSQSVPFSATELLLASAIFCLVFWVLKGLRPRVPKGLKSPPEPWGWPLLGHVLTLGKN
PHLALSRMSQRYGDVLQIRIGSTPVLVLSRLDTIRQALVRQGDDFKGRPDLYTSTLITDG
QSLTFSTDSGPVWAARRRLAQNALNTFSIASDPASSSSCYLEEHVSKEAKALISRLQELM
AGPGHFDPYNQVVVSVANVIGAMCFGQHFPESSDEMLSLVKNTHEFVETASSGNPLDFFP
ILRYLPNPALQRFKAFNQRFLWFLQKTVQEHYQDFDKNSVRDITGALFKHSKKGPRASGN
LIPQEKIVNLVNDIFGAGFDTVTTAISWSLMYLVTKPEIQRKIQKELDTVIGRERRPRLS
DRPQLPYLEAFILETFRHSSFLPFTIPHSTTRDTTLNGFYIPKKCCVFVNQWQVNHDPEL
WEDPSEFRPERFLTADGTAINKPLSEKMMLFGMGKRRCIGEVLAKWEIFLFLAILLQQLE
FSVPPGVKVDLTPIYGLTMKHARCEHVQARLRFSIN
|
|
|
|---|
| BDBM50293855 |
|---|
| n/a |
|---|
| Name | BDBM50293855 |
|---|
| Synonyms: | 4-{3-[(2R)-2,3-dihydroxypropyl]-2-oxo-2,3-dihydro-1H-benzimidazol-1-yl}-1-[(1S,3S,4R)-spiro[bicyclo[2.2.1]heptane-2,1'-cyclopropan]-3-ylmethyl]piperidine hydrochloride | CHEMBL564840 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C25H35N3O3 |
|---|
| Mol. Mass. | 425.5637 |
|---|
| SMILES | OC[C@H](O)Cn1c2ccccc2n(C2CCN(C[C@H]3[C@H]4CC[C@H](C4)C33CC3)CC2)c1=O |r| |
|---|
| Structure | 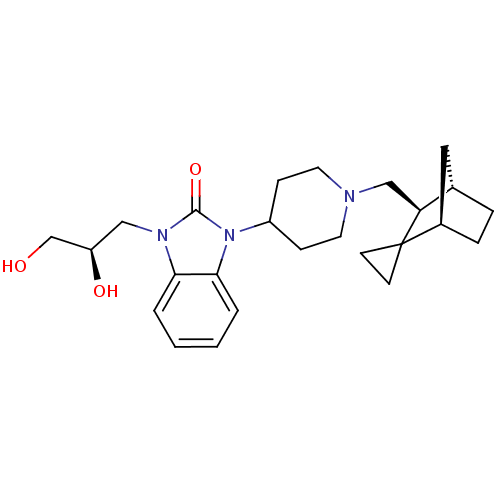
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Satoh, A; Sagara, T; Sakoh, H; Hashimoto, M; Nakashima, H; Kato, T; Goto, Y; Mizutani, S; Azuma-Kanoh, T; Tani, T; Okuda, S; Okamoto, O; Ozaki, S; Iwasawa, Y; Ohta, H; Kawamoto, H Identification of an orally active opioid receptor-like 1 (ORL1) receptor antagonist 4-{3-[(2R)-2,3-dihydroxypropyl]-2-oxo-2,3-dihydro-1H-benzimidazol-1-yl}-1-[(1S,3S,4R)-spiro[bicyclo[2.2.1]heptane-2,1'-cyclopropan]-3-ylmethyl]piperidine as clinical candidate. J Med Chem52:4091-4 (2009) [PubMed] Article
Satoh, A; Sagara, T; Sakoh, H; Hashimoto, M; Nakashima, H; Kato, T; Goto, Y; Mizutani, S; Azuma-Kanoh, T; Tani, T; Okuda, S; Okamoto, O; Ozaki, S; Iwasawa, Y; Ohta, H; Kawamoto, H Identification of an orally active opioid receptor-like 1 (ORL1) receptor antagonist 4-{3-[(2R)-2,3-dihydroxypropyl]-2-oxo-2,3-dihydro-1H-benzimidazol-1-yl}-1-[(1S,3S,4R)-spiro[bicyclo[2.2.1]heptane-2,1'-cyclopropan]-3-ylmethyl]piperidine as clinical candidate. J Med Chem52:4091-4 (2009) [PubMed] Article