

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Reaction Details | |||
|---|---|---|---|
 | Report a problem with these data | ||
| Target | 11-beta-hydroxysteroid dehydrogenase 1 | ||
| Ligand | BDBM50294765 | ||
| Substrate/Competitor | n/a | ||
| Meas. Tech. | ChEMBL_576303 (CHEMBL1030305) | ||
| IC50 | 17±n/a nM | ||
| Citation |  Roche, D; Carniato, D; Leriche, C; Lepifre, F; Christmann-Franck, S; Graedler, U; Charon, C; Bozec, S; Doare, L; Schmidlin, F; Lecomte, M; Valeur, E Discovery and structure-activity relationships of pentanedioic acid diamides as potent inhibitors of 11beta-hydroxysteroid dehydrogenase type I. Bioorg Med Chem Lett19:2674-8 (2009) [PubMed] Article Roche, D; Carniato, D; Leriche, C; Lepifre, F; Christmann-Franck, S; Graedler, U; Charon, C; Bozec, S; Doare, L; Schmidlin, F; Lecomte, M; Valeur, E Discovery and structure-activity relationships of pentanedioic acid diamides as potent inhibitors of 11beta-hydroxysteroid dehydrogenase type I. Bioorg Med Chem Lett19:2674-8 (2009) [PubMed] Article | ||
| More Info.: | Get all data from this article, Assay Method | ||
| 11-beta-hydroxysteroid dehydrogenase 1 | |||
| Name: | 11-beta-hydroxysteroid dehydrogenase 1 | ||
| Synonyms: | 11-DH | 11-beta-HSD1 | 11-beta-Hydroxysteroid Dehydrogenase 1 (11-beta-HSD1) | 11-beta-hydroxysteroid dehydrogenase | 11-beta-hydroxysteroid dehydrogenase 1 (11HSD1) | Corticosteroid 11-beta-dehydrogenase isozyme 1 | Corticosteroid 11-beta-dehydrogenase isozyme 1 (11-beta-HSD-1) | Corticosteroid 11-beta-dehydrogenase isozyme 1 (11-beta-HSD1) | Corticosteroid 11-beta-dehydrogenase isozyme 1 (11beta-HSD1) | DHI1_HUMAN | HSD11 | HSD11B1 | HSD11L | SDR26C1 | ||
| Type: | Enzyme | ||
| Mol. Mass.: | 32409.16 | ||
| Organism: | Homo sapiens (Human) | ||
| Description: | P28845 | ||
| Residue: | 292 | ||
| Sequence: |
| ||
| BDBM50294765 | |||
| n/a | |||
| Name | BDBM50294765 | ||
| Synonyms: | CHEMBL551306 | N-Adamantan-2-yl-2-{1-[(4-fluoro-phenylcarbamoyl)-methyl]-cyclopentyl}-acetamide | ||
| Type | Small organic molecule | ||
| Emp. Form. | C25H33FN2O2 | ||
| Mol. Mass. | 412.5401 | ||
| SMILES | Fc1ccc(NC(=O)CC2(CC(=O)NC3C4CC5CC(C4)CC3C5)CCCC2)cc1 |TLB:20:19:23:16.15.14,20:15:18.19.21:23,THB:14:15:18:21.22.23,14:22:18:16.20.15,13:14:18.19.21:23,(8.39,2.65,;7.05,3.42,;5.72,2.65,;4.39,3.41,;4.39,4.96,;3.05,5.73,;1.72,4.96,;1.72,3.42,;.38,5.72,;-.95,4.95,;-2.28,5.72,;-3.61,4.94,;-3.61,3.4,;-4.95,5.71,;-6.23,4.85,;-6.24,3.33,;-7.64,2.98,;-8.97,3.46,;-10.16,2.19,;-8.66,2.61,;-7.26,2.05,;-8.67,4.2,;-7.63,5.43,;-8.98,4.95,;-2.19,4.03,;-1.7,2.57,;-.16,2.59,;.31,4.06,;5.71,5.73,;7.04,4.97,)| | ||
| Structure | 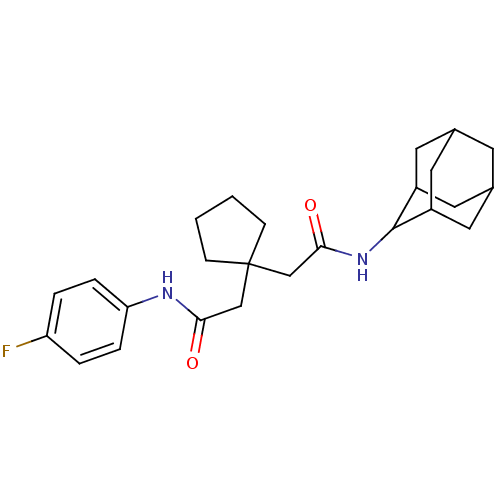
| ||