| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Fatty-acid amide hydrolase 1 |
|---|
| Ligand | BDBM50296484 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_581830 (CHEMBL1064166) |
|---|
| IC50 | 78±n/a nM |
|---|
| Citation |  Hart, T; Macias, AT; Benwell, K; Brooks, T; D'Alessandro, J; Dokurno, P; Francis, G; Gibbons, B; Haymes, T; Kennett, G; Lightowler, S; Mansell, H; Matassova, N; Misra, A; Padfield, A; Parsons, R; Pratt, R; Robertson, A; Walls, S; Wong, M; Roughley, S Fatty acid amide hydrolase inhibitors. Surprising selectivity of chiral azetidine ureas. Bioorg Med Chem Lett19:4241-4 (2009) [PubMed] Article Hart, T; Macias, AT; Benwell, K; Brooks, T; D'Alessandro, J; Dokurno, P; Francis, G; Gibbons, B; Haymes, T; Kennett, G; Lightowler, S; Mansell, H; Matassova, N; Misra, A; Padfield, A; Parsons, R; Pratt, R; Robertson, A; Walls, S; Wong, M; Roughley, S Fatty acid amide hydrolase inhibitors. Surprising selectivity of chiral azetidine ureas. Bioorg Med Chem Lett19:4241-4 (2009) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Fatty-acid amide hydrolase 1 |
|---|
| Name: | Fatty-acid amide hydrolase 1 |
|---|
| Synonyms: | Anandamide amidohydrolase | Anandamide amidohydrolase 1 | FAAH | FAAH1 | FAAH1_HUMAN | Fatty Acid Amide Hydrolase (FAAH) | Fatty-acid amide hydrolase (FAAH) | Fatty-acid amide hydrolase 1 | Oleamide hydrolase 1 |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 63071.19 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | O00519 |
|---|
| Residue: | 579 |
|---|
| Sequence: | MVQYELWAALPGASGVALACCFVAAAVALRWSGRRTARGAVVRARQRQRAGLENMDRAAQ
RFRLQNPDLDSEALLALPLPQLVQKLHSRELAPEAVLFTYVGKAWEVNKGTNCVTSYLAD
CETQLSQAPRQGLLYGVPVSLKECFTYKGQDSTLGLSLNEGVPAECDSVVVHVLKLQGAV
PFVHTNVPQSMFSYDCSNPLFGQTVNPWKSSKSPGGSSGGEGALIGSGGSPLGLGTDIGG
SIRFPSSFCGICGLKPTGNRLSKSGLKGCVYGQEAVRLSVGPMARDVESLALCLRALLCE
DMFRLDPTVPPLPFREEVYTSSQPLRVGYYETDNYTMPSPAMRRAVLETKQSLEAAGHTL
VPFLPSNIPHALETLSTGGLFSDGGHTFLQNFKGDFVDPCLGDLVSILKLPQWLKGLLAF
LVKPLLPRLSAFLSNMKSRSAGKLWELQHEIEVYRKTVIAQWRALDLDVVLTPMLAPALD
LNAPGRATGAVSYTMLYNCLDFPAGVVPVTTVTAEDEAQMEHYRGYFGDIWDKMLQKGMK
KSVGLPVAVQCVALPWQEELCLRFMREVERLMTPEKQSS
|
|
|
|---|
| BDBM50296484 |
|---|
| n/a |
|---|
| Name | BDBM50296484 |
|---|
| Synonyms: | (S)-{3-[(4-Chlorophenyl)-(2-chloropyridin-3-yl)methoxy]azetidin-1-yl}piperidin-1-ylmethanone | CHEMBL555563 | VER-156084 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C21H23Cl2N3O2 |
|---|
| Mol. Mass. | 420.332 |
|---|
| SMILES | Clc1ccc(cc1)[C@H](OC1CN(C1)C(=O)N1CCCCC1)c1cccnc1Cl |r| |
|---|
| Structure | 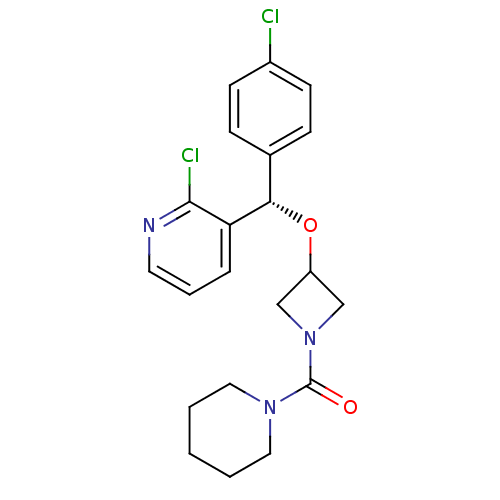
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Hart, T; Macias, AT; Benwell, K; Brooks, T; D'Alessandro, J; Dokurno, P; Francis, G; Gibbons, B; Haymes, T; Kennett, G; Lightowler, S; Mansell, H; Matassova, N; Misra, A; Padfield, A; Parsons, R; Pratt, R; Robertson, A; Walls, S; Wong, M; Roughley, S Fatty acid amide hydrolase inhibitors. Surprising selectivity of chiral azetidine ureas. Bioorg Med Chem Lett19:4241-4 (2009) [PubMed] Article
Hart, T; Macias, AT; Benwell, K; Brooks, T; D'Alessandro, J; Dokurno, P; Francis, G; Gibbons, B; Haymes, T; Kennett, G; Lightowler, S; Mansell, H; Matassova, N; Misra, A; Padfield, A; Parsons, R; Pratt, R; Robertson, A; Walls, S; Wong, M; Roughley, S Fatty acid amide hydrolase inhibitors. Surprising selectivity of chiral azetidine ureas. Bioorg Med Chem Lett19:4241-4 (2009) [PubMed] Article