| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cannabinoid receptor 1 |
|---|
| Ligand | BDBM50301747 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_597914 (CHEMBL1042699) |
|---|
| Ki | 0.1±n/a nM |
|---|
| Citation |  Dow, RL; Hadcock, JR; Scott, DO; Schneider, SR; Paight, ES; Iredale, PA; Carpino, PA; Griffith, DA; Hammond, M; Dasilva-Jardine, P Bioisosteric replacement of the hydrazide pharmacophore of the cannabinoid-1 receptor antagonist SR141716A. Part I: potent, orally-active 1,4-disubstituted imidazoles. Bioorg Med Chem Lett19:5351-4 (2009) [PubMed] Article Dow, RL; Hadcock, JR; Scott, DO; Schneider, SR; Paight, ES; Iredale, PA; Carpino, PA; Griffith, DA; Hammond, M; Dasilva-Jardine, P Bioisosteric replacement of the hydrazide pharmacophore of the cannabinoid-1 receptor antagonist SR141716A. Part I: potent, orally-active 1,4-disubstituted imidazoles. Bioorg Med Chem Lett19:5351-4 (2009) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cannabinoid receptor 1 |
|---|
| Name: | Cannabinoid receptor 1 |
|---|
| Synonyms: | CANN6 | CANNABINOID CB1 | CB-R | CB1 | CNR | CNR1 | CNR1_HUMAN | Cannabinoid CB1 receptor | Cannabinoid receptor | Cannabinoid receptor 1 (CB1) | Cannabinoid receptor 1 (brain) |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 52868.96 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P21554 |
|---|
| Residue: | 472 |
|---|
| Sequence: | MKSILDGLADTTFRTITTDLLYVGSNDIQYEDIKGDMASKLGYFPQKFPLTSFRGSPFQE
KMTAGDNPQLVPADQVNITEFYNKSLSSFKENEENIQCGENFMDIECFMVLNPSQQLAIA
VLSLTLGTFTVLENLLVLCVILHSRSLRCRPSYHFIGSLAVADLLGSVIFVYSFIDFHVF
HRKDSRNVFLFKLGGVTASFTASVGSLFLTAIDRYISIHRPLAYKRIVTRPKAVVAFCLM
WTIAIVIAVLPLLGWNCEKLQSVCSDIFPHIDETYLMFWIGVTSVLLLFIVYAYMYILWK
AHSHAVRMIQRGTQKSIIIHTSEDGKVQVTRPDQARMDIRLAKTLVLILVVLIICWGPLL
AIMVYDVFGKMNKLIKTVFAFCSMLCLLNSTVNPIIYALRSKDLRHAFRSMFPSCEGTAQ
PLDNSMGDSDCLHKHANNAASVHRAAESCIKSTVKIAKVTMSVSTDTSAEAL
|
|
|
|---|
| BDBM50301747 |
|---|
| n/a |
|---|
| Name | BDBM50301747 |
|---|
| Synonyms: | 1-(2-chlorophenyl)-5-(4-chlorophenyl)-4-methyl-3-(1-(2-phenylpropan-2-yl)-1H-imidazol-4-yl)-1H-pyrazole | CHEMBL578969 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C28H24Cl2N4 |
|---|
| Mol. Mass. | 487.423 |
|---|
| SMILES | Cc1c(nn(c1-c1ccc(Cl)cc1)-c1ccccc1Cl)-c1cn(cn1)C(C)(C)c1ccccc1 |
|---|
| Structure | 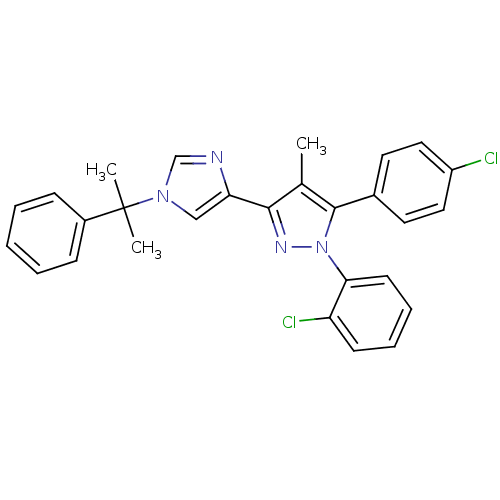
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Dow, RL; Hadcock, JR; Scott, DO; Schneider, SR; Paight, ES; Iredale, PA; Carpino, PA; Griffith, DA; Hammond, M; Dasilva-Jardine, P Bioisosteric replacement of the hydrazide pharmacophore of the cannabinoid-1 receptor antagonist SR141716A. Part I: potent, orally-active 1,4-disubstituted imidazoles. Bioorg Med Chem Lett19:5351-4 (2009) [PubMed] Article
Dow, RL; Hadcock, JR; Scott, DO; Schneider, SR; Paight, ES; Iredale, PA; Carpino, PA; Griffith, DA; Hammond, M; Dasilva-Jardine, P Bioisosteric replacement of the hydrazide pharmacophore of the cannabinoid-1 receptor antagonist SR141716A. Part I: potent, orally-active 1,4-disubstituted imidazoles. Bioorg Med Chem Lett19:5351-4 (2009) [PubMed] Article